Andrew J. Bradley M.D.1; Scott P. Baute, PA-C1; Stephen P. Robie, PA-C1; Robert K. Zeman, M.D.2; Andrew D. Choi, M.D.1,2
1Division of Cardiology, The George Washington University School of Medicine
2Department of Radiology, The George Washington University School of Medicine, Washington, D.C., U.S.A.
Clinical History:
A 55 year old previously healthy woman was evaluated for progressive dyspnea on exertion with associated chest tightness. She denied fevers, chills, and night sweats. She underwent a 2D-Echocardiogram which showed a large pericardial effusion with thickening of the right atrial wall and a mass just below the posterior tricuspid leaflet (Movie 1). Left and right ventricular systolic function was normal. Pericardiocentesis yielded 850 mL of hemorrhagic fluid, analysis of which was nondiagnostic. A CT of the chest, abdomen, and pelvis demonstrated numerous lung nodules, ground glass lesions and diffuse mediastinal lymphadenopathy (Image 1). In addition, the cardiac mass was visualized. To define the probable etiology of the mass, cardiac MRI was performed for more precise localization and tissue characterization.
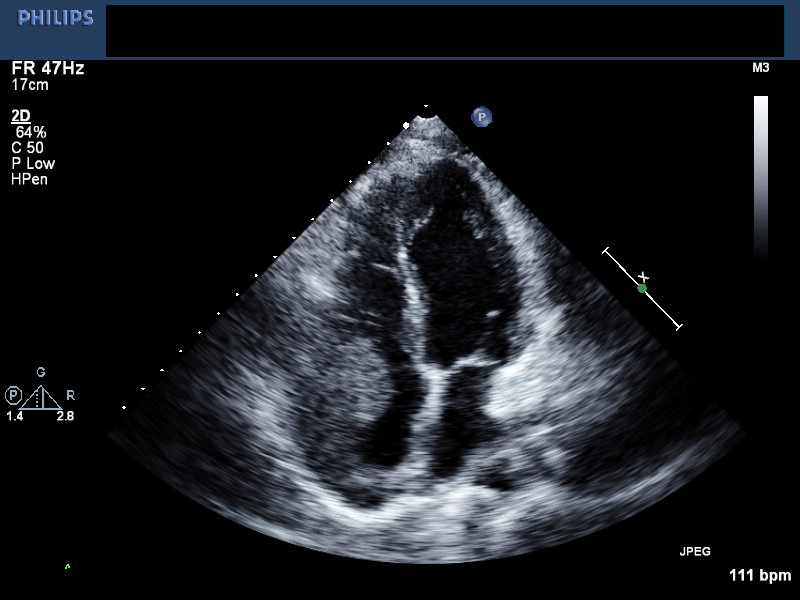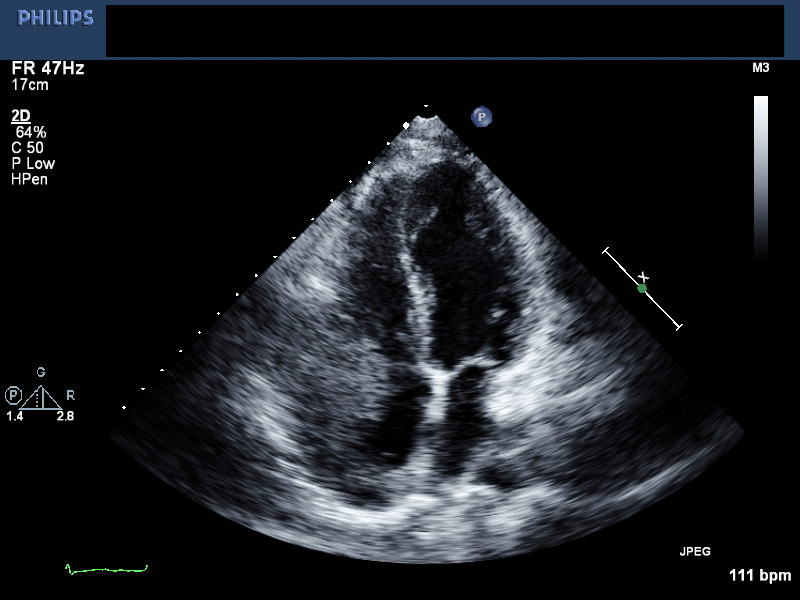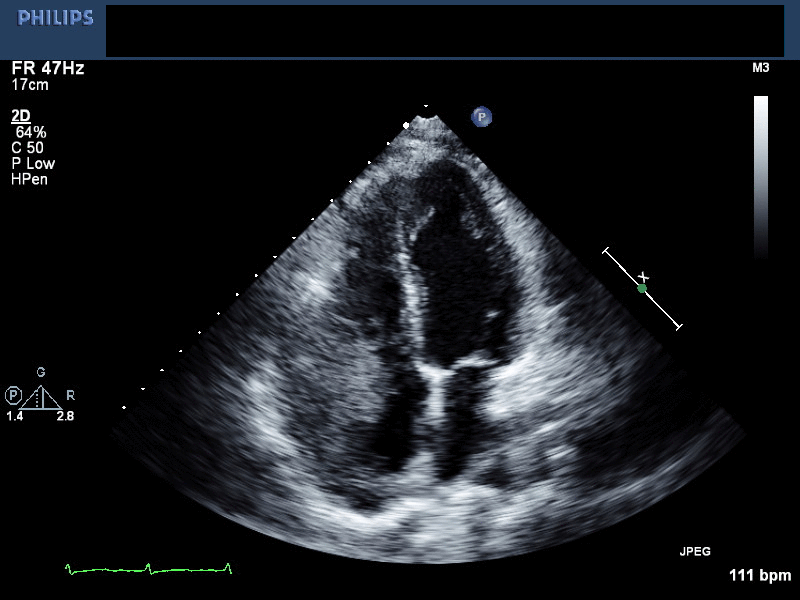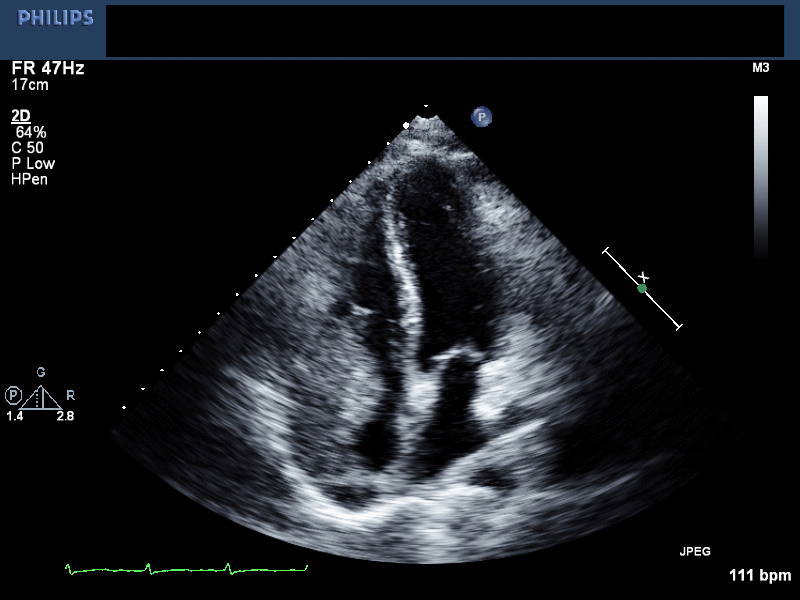
Movie 1. 2D-Echocardiogram in the apical 4 chamber view showing mass in the right ventricle and right atrium. The effusion has been successfully drained.
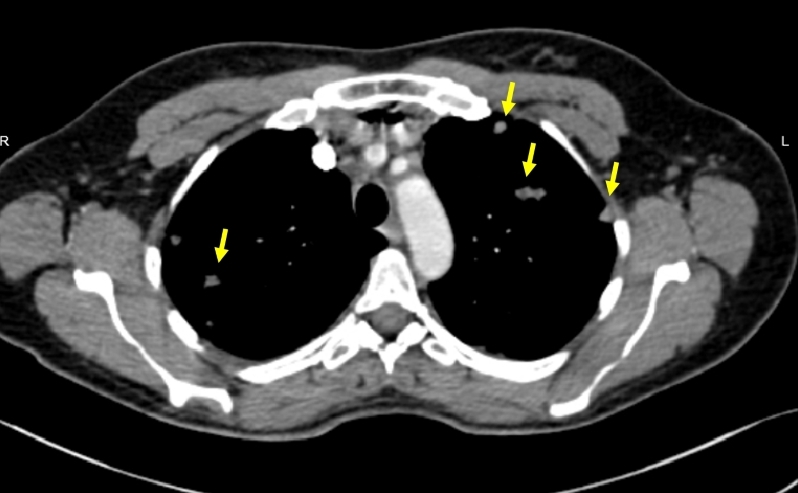
Image 1. Representative Chest-CT at the level of the aortic arch. Yellow arrows point to pulmonary nodules.
Cardiac MRI Findings:
Cardiac MRI was performed with a 1.5 T MRI system (Discovery MR450, General Electric, Wisconsin, USA). Steady State Bright Blood FIESTA cine demonstrated a markedly abnormal cardiac morphology with a large 10 cm x 4 cm soft tissue mass involving the anterior portions of both the right atrium and right ventricle (Movie 2-5). The mass extended into the AV groove from the level of the main pulmonary artery to the inferior surface of the right ventricle. There was infiltration of the roof of the right ventricle that extended posteriorly encasing the anterior surface of the aortic root. The right coronary artery appeared to enhance, but was markedly attenuated as it passed anteriorly directly into the mass indicating encasement of this vessel (Movie 6).
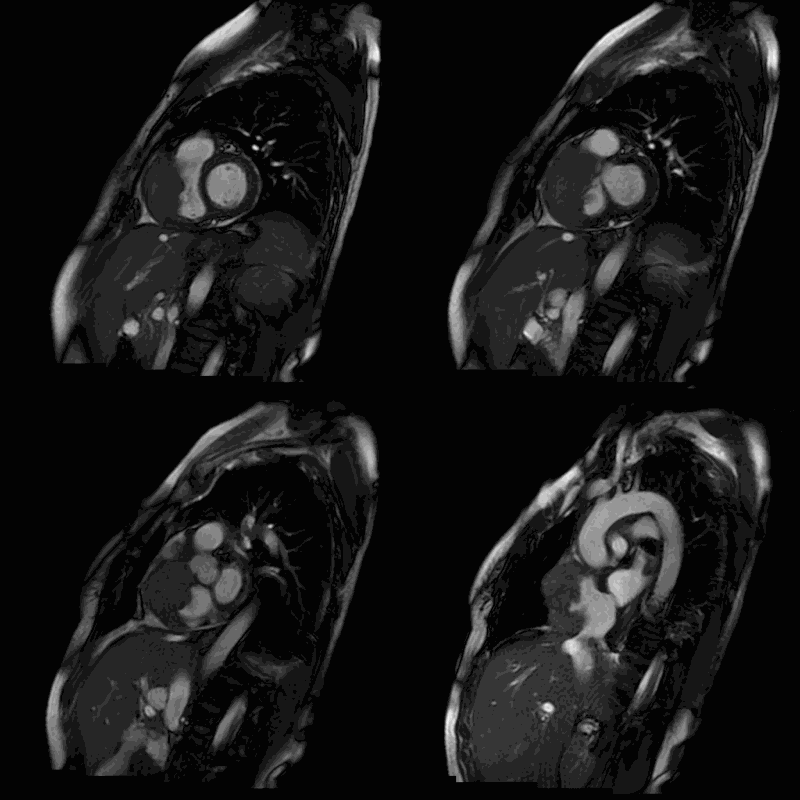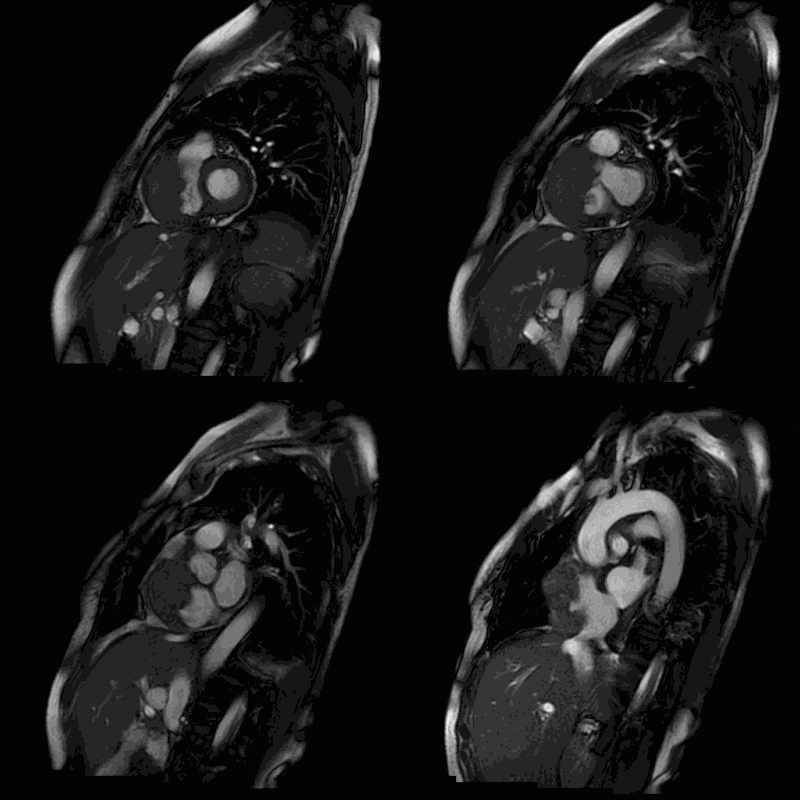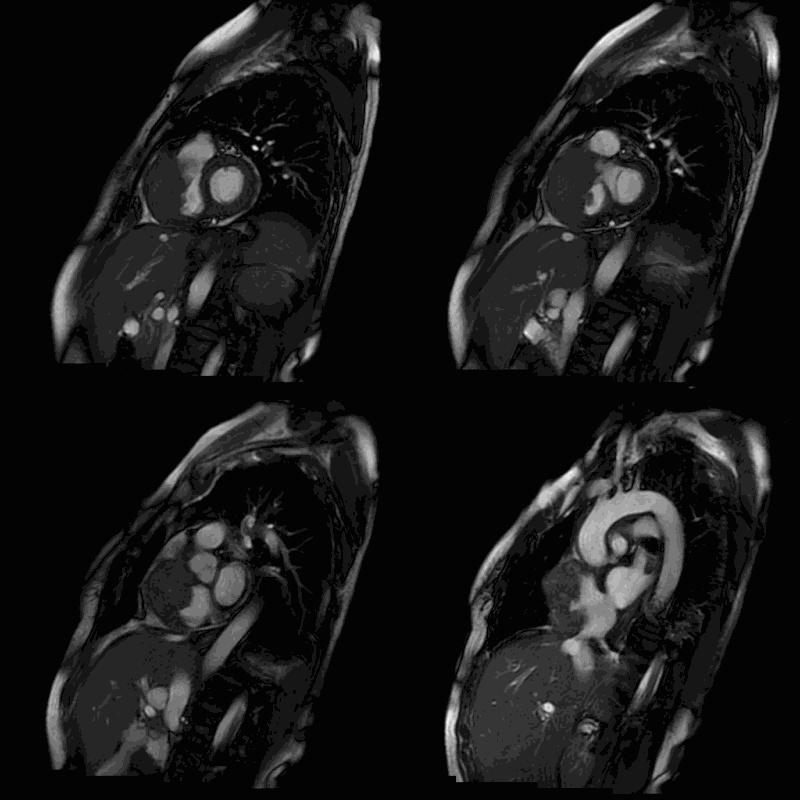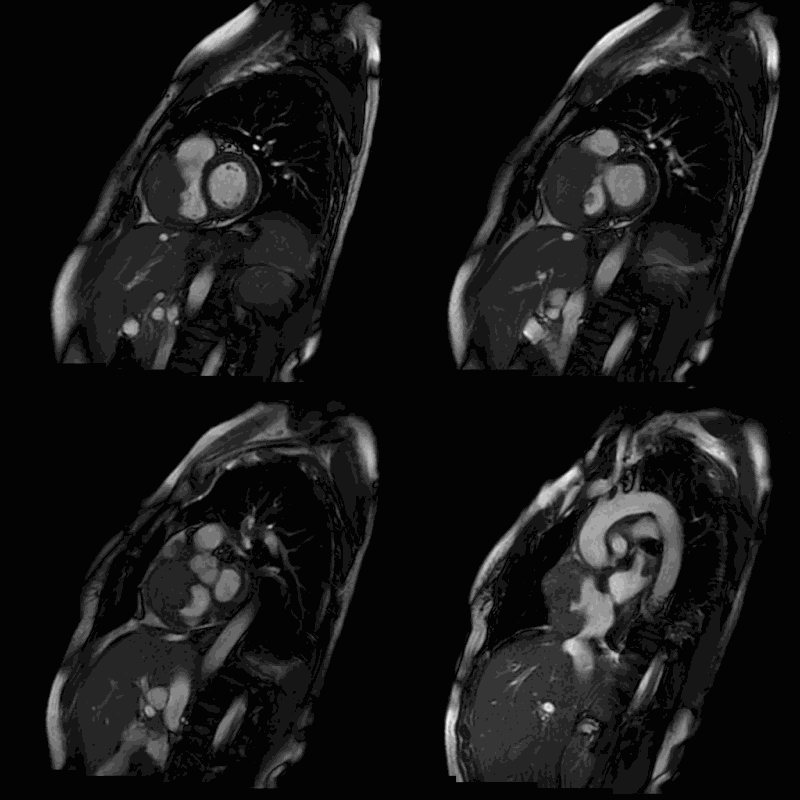
Movie 2-5. Cine steady state bright blood FIESTA short axis stack showing a large mass of the anterior wall of the right atrium and ventricle.
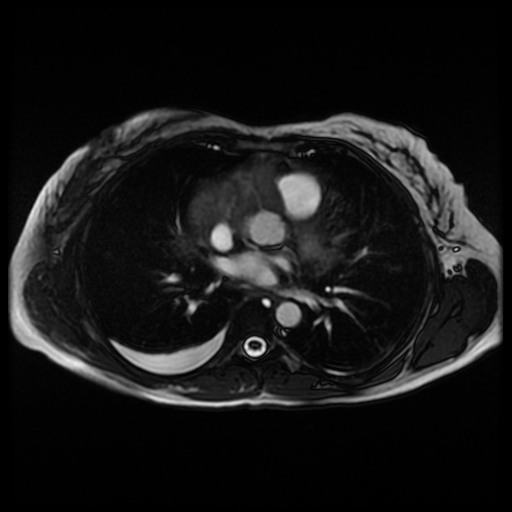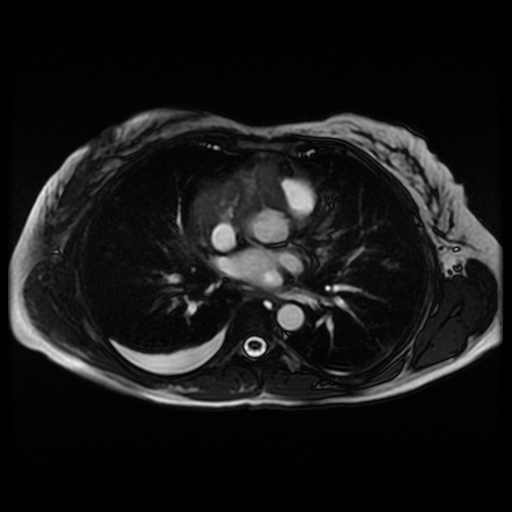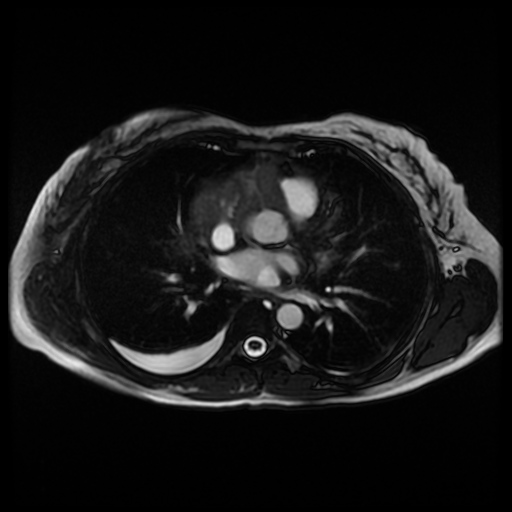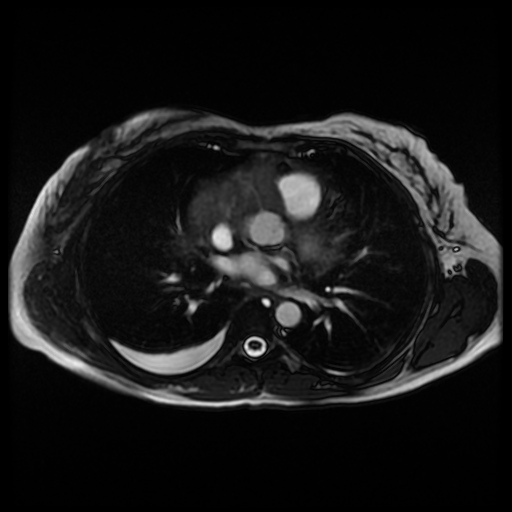
Movie 6. Cine steady state bright blood FIESTA axial showing a mass encasing the right coronary artery.
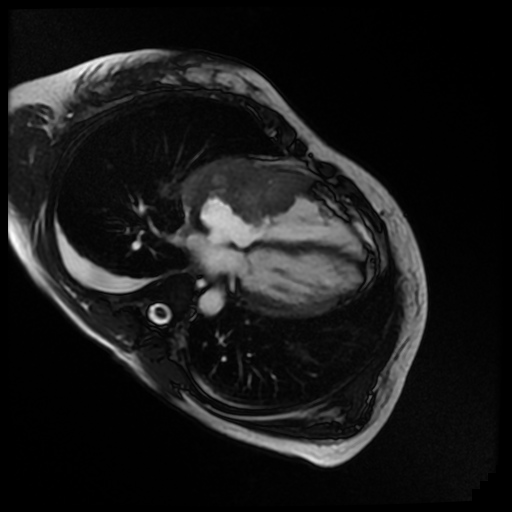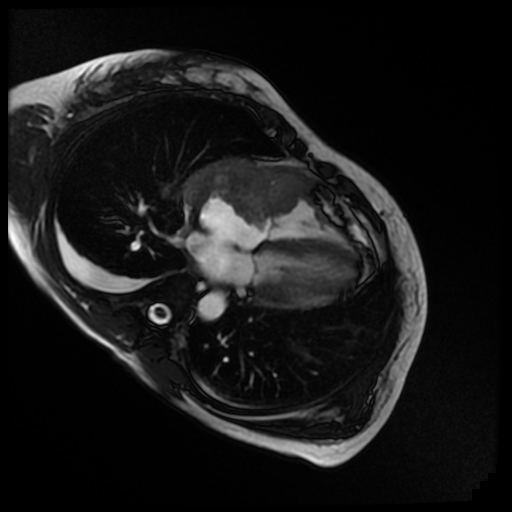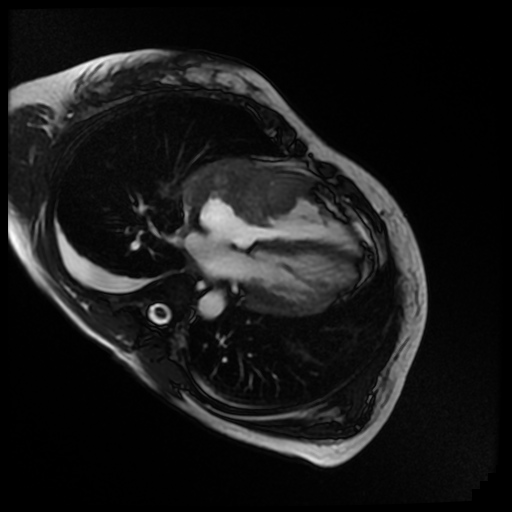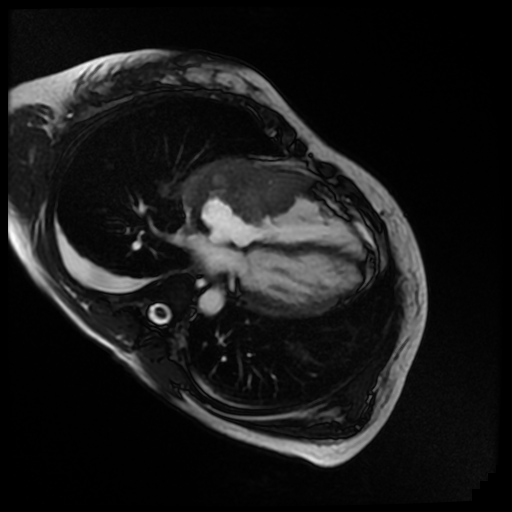
Movie 7. Cine steady state bright blood FIESTA four chamber showing the mass of the anterior wall of the right atrium and ventricle with mobility into the tricuspid valve.
On T2-weighted black blood (“Double IR”) imaging demonstrated increased signal indicative of fluid or vascular components of the tumor (Image 2). On Double IR imaging with fat saturation and T1-weighted fast-spin echo (“Triple IR”) imaging, there was no evidence of fatty infiltration (Image 3-4). On gadolinium (gadopentetate dimeglumine, “Magnevist”, Bayer HealthCare, NJ, USA) first pass perfusion imaging, there were some areas of contrast uptake within the mid-portion of the mass, suggestive of a vascular component (Movie 8, Image 5-6). Also noted were contrast enhancement pulmonary nodules. Late gadolinium enhanced images showed heterogeneous enhancement throughout the mass (Image 7).
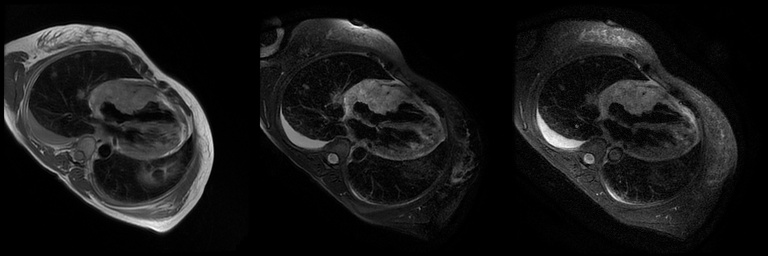
Image 2-4. T2 Weighted double inversion recovery (DIR) without and with fat saturation and T1 weighted triple inversion recovery (TIR) four chamber images respectively. The DIR image shows increased signal indicative of fluid or vascular components of the mass. The DIR with fat saturation and TIR images show no evidence of fatty infiltration of the mass.

Movie 8 and Image 5-6. First pass perfusion coronal shows uptake of gadolinium within the mid-portion of the mass (yellow arrows).
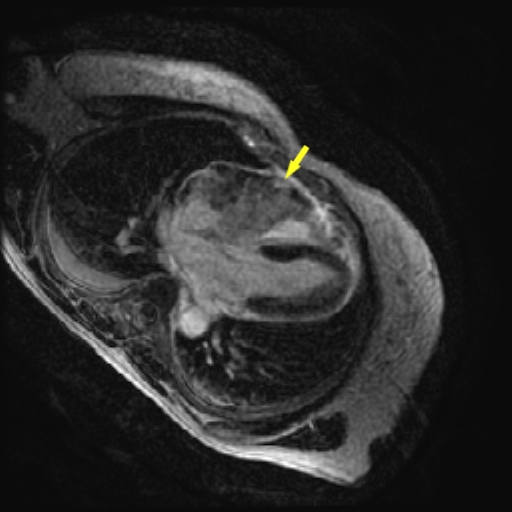
Image 7. Late gadolinium enhancement in the axial plane demonstrating areas of enhancement within the angiosarcoma in the right ventricle (yellow arrow). There is no late gadolinium enhancement in the left ventricular myocardium.
Conclusion:
The cardiac MRI findings in combination with the clinical history were highly suggestive of angiosarcoma. A mini-thoracotomy was performed for surgical biopsy of the mass with a concomitant lung nodule biopsy that confirmed the diagnosis of angiosarcoma. The patient was referred to begin outpatient chemotherapy.
Perspective
In this case, we present a patient with primary angiosarcoma of the myocardium. Cardiac MRI enabled localization and tissue characterization of the mass that was subsequently confirmed on myocardial biopsy.
Angiosarcomas predominately affect middle-aged males and typically arise from the right atrium near the atrioventricular groove, though given their rare nature, the exact epidemiology is unclear (1). Symptoms at time of clinical presentation are often vague unless there is obstruction of neighboring structures which is dependent on the location of the tumor. As a result, angiosarcomas are frequently diagnosed late on the basis of their metastasis to the lung, lymph nodes, and liver (2).
Transthoracic echocardiography serves as the initial test to identify cardiac tumors. Further study with cardiac MRI allows for the differentiation between malignant and benign lesions. In angiosarcoma, areas of vascularity as well as necrosis are seen. It is important to note that late gadolinium enhancement is not specific for necrosis and may also be seen with fibrosis. While contrast echocardiography has been studied as a method to characterize masses as either benign or malignant, MRI has the added benefits of determining extent of infiltration into other cardiac chambers and vascular structures and evaluating for metastasis to the mediastinum, pleura, liver, and to some extent lungs (3, 4, 5). A case series of 12 patients has been reported discussing the value of MRI in differentiating cardiac angiosarcoma from lymphoma. In that series, all seven of the lymphomas completely encased the right coronary artery as compared to none of the angiosarcomas (6). Our patient’s angiosarcoma, in comparison did completely surround the right coronary artery.
Therapeutically, resection of angiosarcoma is possible but is difficult given the tumor’s infiltrative nature and that it is typically already in an advanced stage at presentation. In our patient, MRI demonstrated encasement of the right coronary artery which would greatly hinder efforts at resection. Surgical approaches generally focus on debulking with adjuvant chemotherapy and radiation. Despite these potential interventions, prognosis of angiosarcoma is exceedingly poor (1).
A novel technique from a single center, cardiac autotransplantation for left sided primary cardiac tumors, has been shown to increase the length of survival when compared to standard cardiac sarcoma resection. The operative technique involves near-complete explantation of the heart (the left atrium is divided anterior to the pulmonary veins) and then resection of the tumor with the heart immersed in an ice bath. Surrounding structures in the chest are also assessed for tumor burden with disease resected as needed (5, 6). A referral for cardiac autotransplantation was strongly considered for this patient. However, cardiac MRI findings were the crucial determinant regarding the extent of infiltration that made this option unfeasible.
In conclusion, cardiac MRI is the preferred imaging technique for the evaluation of myocardial masses and can direct both diagnosis and therapy, though treatment options for malignancy are often limited at the time of diagnosis.
Click here to view case on CloudCMR
References:
(1) Burke A, Jeudy J, Virmani R. Cardiac Tumours: an update. Heart. 2008;94:117-123
Case prepared by Jason Johnson, MD MHS, Associate Editor, SCMR Case of the Week







