Institute: Emory University, Atlanta, Georgia
Clinical Presentation
A 57 year old woman with a history of sinus venosus atrial septal defect and partial anomalous pulmonary venous return (PAPVR) repaired in childhood presented to the adult congenital heart disease clinic with junctional rhythm and significant lower extremity swelling. She had three surgeries during childhood for congenital heart disease. She did not have consistent care for her heart disease and her surgical history was not clear at presentation. In addition to her congenital heart disease she had known active hepatitis C infection. A month before presentation, she was evaluated for dizziness with bradycardia. She continued to have intermittent bouts of dizziness without syncope. On physical exam, she had significant lower body edema. She was unable to control her lower extremity edema with a torsemide and metolazone combination. Her ECG in clinic showed a junctional rhythm and right ventricular hypertrophy. (Figure 1) During her admission, an echocardiogram revealed normal biventricular systolic function. She underwent a CT of the chest which showed a dilated hemiazygous and inferior vena cava. (IVC) (Figure 2)
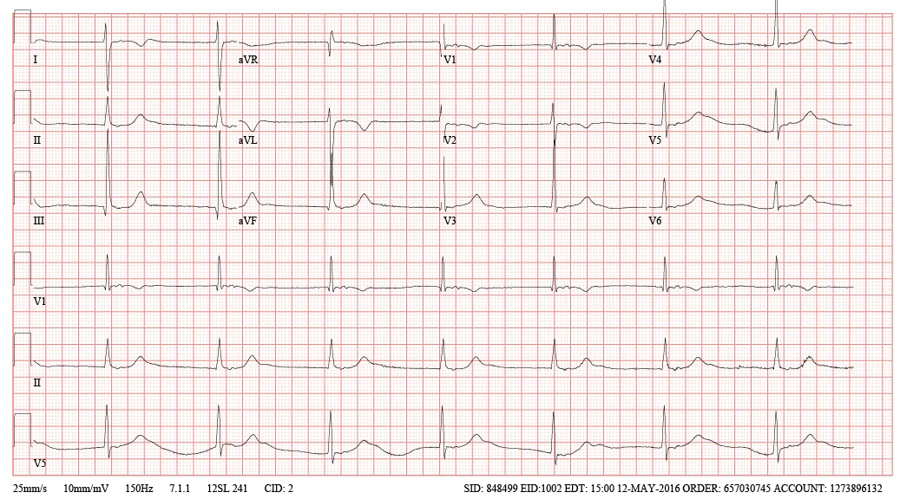
Figure 1
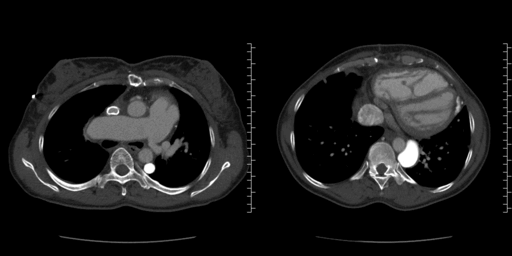
Figure 2A and Figure 2B
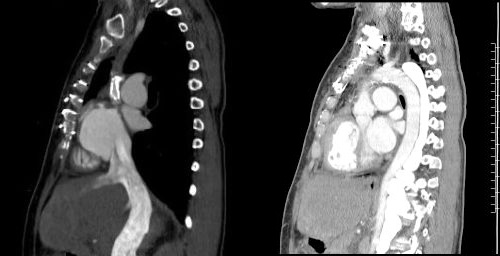
Figure 2C and Figure 2D
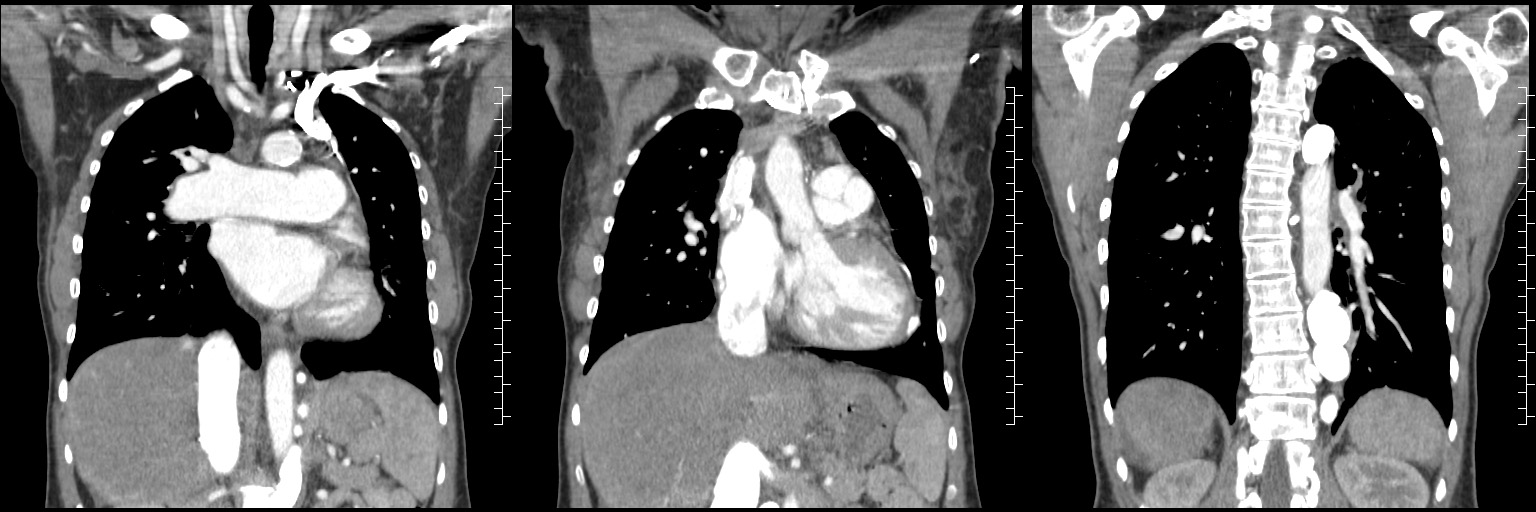
Figure 2E, Figure 2F, and Figure 2G
Figure 2: CT angiography of the chest with contrast. A: Axial views show calcification of prior surgical repair of the superior vena cava (SVC). The left pulmonary artery has severe stenosis and there is contrast in the hemiazygous. B: Dilated hemiazygous with contrast and a dilated IVC. C. Bicaval view showing patent IVC and calcified SVC. Contrast is starting to opacify the IVC. D. Sagittal view of the aortic arch and the significantly dilated hemiazygous which is filled with contrast. E-G. Contrast was injected into the left arm and fills the hemiazygous. The SVC is not opacified with contrast.
She was referred for cardiac MRI to better evaluate her PAPVR repair and sinus venous repair.
CMR Findings
CMR imaging revealed patent drainage of the anomalous right upper pulmonary vein into the left atrium through a baffle repair. There is no evidence of a residual atrial septal defect. The SVC had complete obstruction above the level of the repair. A large hemiazygous vein drains into the infra-hepatic IVC. The left pulmonary artery is diminutive and measures 0.8 cm x 0.7 cm. The right pulmonary artery is dilated. The left ventricular cavity size and systolic function are normal with a calculated left ventricle ejection fraction of 73%. The right ventricle was mildly dilated (94 mL/m2) with normal systolic function. The calculated right ventricular ejection fraction was 55%.
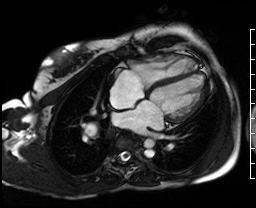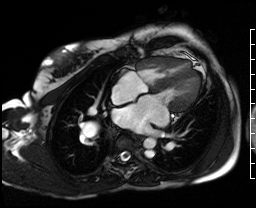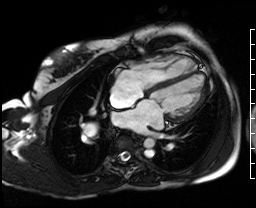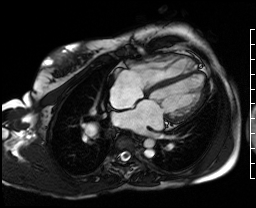
Video Clip 1: A cine horizontal long axis view showing mild right ventricular enlargement.
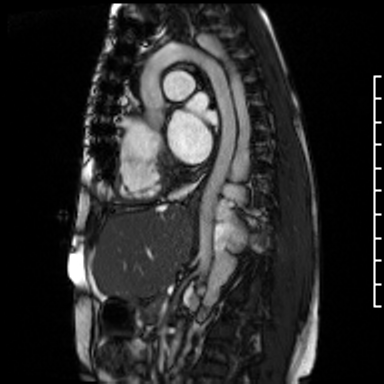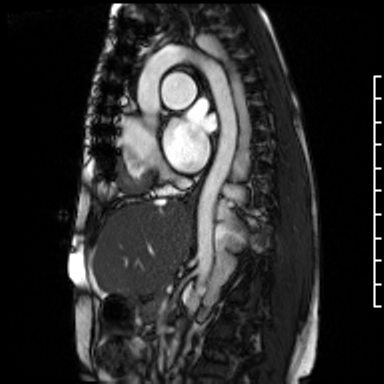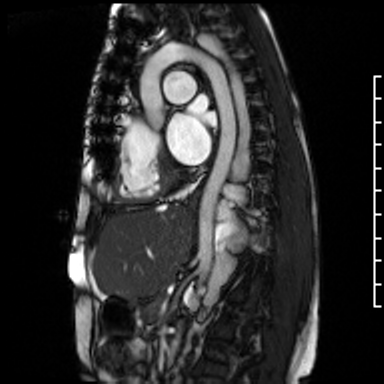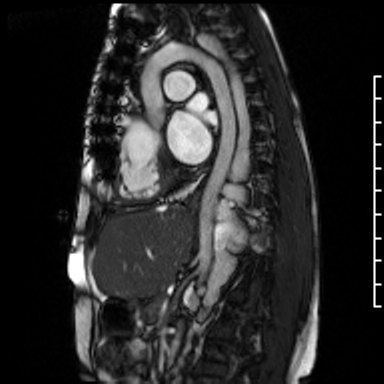
Video Clip 2. Sagittal oblique cine of the aortic arch with the dilated hemiazygous directly posterior to the aorta. The image position was created from a SSFP respiratory navigated 3D dataset.
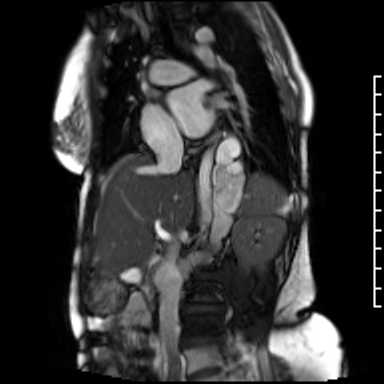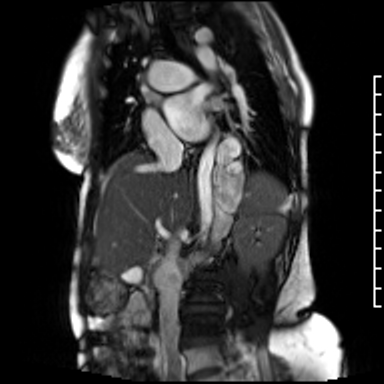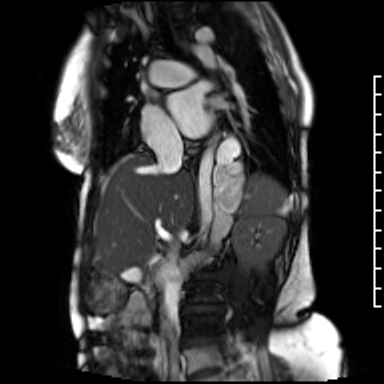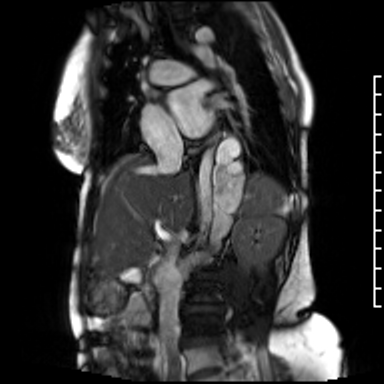
Video Clip 3: Oblique image cine of the hemiazygous draining into the inferior vena cava.
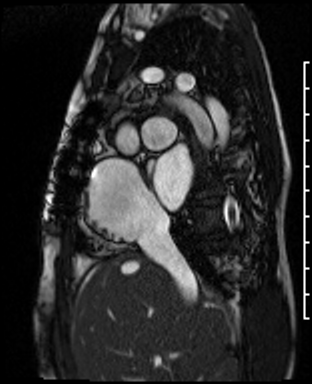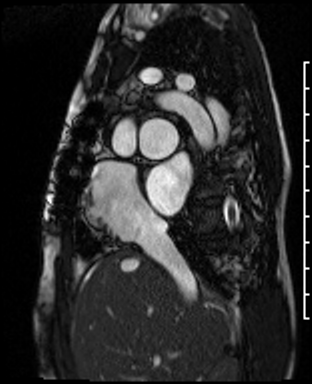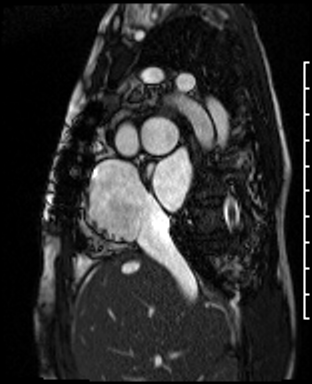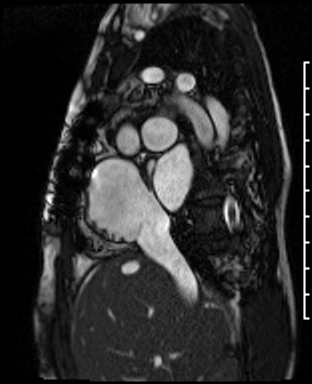
Video Clip 4: Atrial short axis cine stack showed obstruction of the SVC but a patent IVC. There was no residual atrial septal defect.
Flow velocity mapping on phase contrast imaging demonstrated a QP: QS of 1.1:1 that confirmed the absence of significant residual shunt. The MRA showed SVC obstruction above the level of the repair. Contrast opacification from the right arm injection flows into the right subclavian to the left innominate vein and finally drained into a large hemiazygous vein. The hemiazygous vein drained into the infra-hepatic IVC. There is no evidence of IVC obstruction, however the hemiazygous anastomosis to the IVC does appear small. (Figure 2.) There are numerous venous collaterals draining the chest. (Video 5)
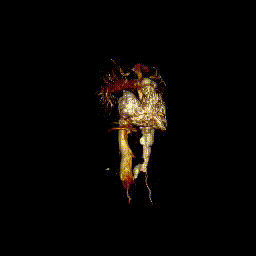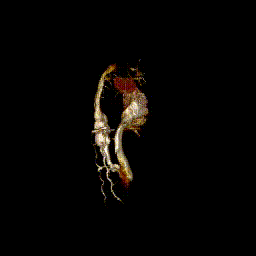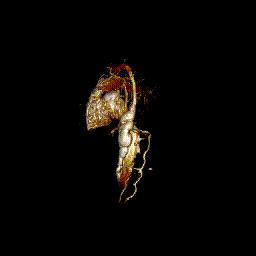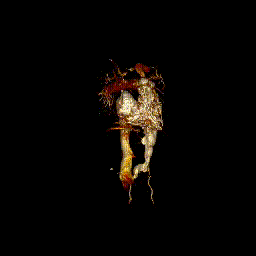
Video clip 5: 3D reconstruction of the venous system.
Delayed enhancement imaging did not show any focal areas of enhancement to suggest scar/fibrosis in the left or right ventricle. The delayed enhancement was repeated with the inversion time set at 600 did not show any thrombus in the dilated hemiazygous.
She also underwent an abdominal and pelvic MRA which did not show IVC obstruction throughout its course from the right atrium to the iliac veins.
The patient was referred for a cardiac catheterization but has been non-compliant with scheduling this.
Conclusion
After anomalous pulmonary venous return repair, it is important to evaluate for both pulmonary venous obstruction and systemic venous obstruction. In our case, the MRA illustrated the drainage pattern of the SVC. Although lower extremity edema is an unusual presentation for SVC obstruction, the patient developed a “pop off” by draining through the hemiazygous and eventually into the IVC. This flow pattern is concerning for a proximal SVC obstruction. Typically, the IVC can tolerate the increased flow and does not cause lower extremity edema. The left pulmonary artery stenosis may increase resistance in the pulmonary circuit and contribute to elevated right atrial pressures. SVC obstruction complicates placement of pacemaker. The short axis cine’s through the atria and the horizontal long axis stack interrogated the patency of the right pulmonary vein and the sinus venous atrial septal defect repair. The QP:QS confirmed there was no residual baffle leak.
Perspective
Partial anomalous pulmonary venous return (PAPVR) occurs in 0.1 – 0.7% of the general population and usually affects the right sided pulmonary veins. PAPVR is typically associated with a sinus venous atrial septal defect.1 If there are multiple anomalous pulmonary veins draining into the right atrium, the veins can cause right sided volume overload and is an indication for repair.2 Diagnosis can occur in either childhood or adulthood. Adults may present with right ventricular enlargement or may be found incidentally on advanced cardiac imaging including cardiac MRI.
The goal of partial anomalous venous return repair is to divert the pulmonary veins back to the left atrium and systemic veins to the right atrium. The most important part of surgical planning is the identification of which pulmonary veins are involved and the presence and location of an atrial septal defect. Most patients have anomalous pulmonary venous return to the superior vena cava. There are three described techniques for right sided pulmonary veins including a single patch technique, double patch technique and the Warden procedure. The single patch technique baffles anomalous pulmonary veins to the left atrium while closing a sinus venous atrial septal defect. The double patch technique using a second patch to enlarge the superior vena cava. Finally, the Warden technique divides superior vena cava above the anomalous pulmonary veins and reimplants the superior vena cava into the right atrium.4
Long term complications after anomalous pulmonary venous return repair include SVC stenosis, pulmonary vein obstruction, sinus node dysfunction or atrial arrhythmias. Said et al reported that SVC obstruction was most common following the Warden procedure, but the Warden procedure was unlikely to cause pulmonary vein stenosis.5 Sonavance et al describes four types of SVC obstruction (partial obstruction, above the azygous arch, below the azygous arch and at the azygous arch) and the how the blood flow is redirected.6 Our patient has a SVC obstruction at the azygous arch, with upper body venous return via the hemiazygous vein to the inferior vena cava.
Click here to view all images on CloudCMR.
References
1. Sormani P, Roghi A, Cereda A et. al. Partial Anomalous Pulmonary Venous Return as Rare Cause of Right Ventricular Dilation: A Retrospective Analysis. Congenit Heart Dis. 2016; 11: 365–8.
2. Fragata J, Magalhaes M, Baquero L, Trigo C, Pinto F, and Fragata I. Partial Anomalous Pulmonary Venous Connections: Surgical Management. World J Pediatr Congenit Heart Surg. 2013 Jan; 4 (1): 44-9.
3. Majdalany DS, Phillis SD, Dearani JA, Connolly HM and Warnes CA. Isolated partial anomalous pulmonary venous connections in adults: Twenty-Year Experience. Congenit Heart Dis. 2010; 5: 537-45.
4. Backer CL and Mavroudis C. “Atrial septal defect, partial anomalous pulmonary venous connection and scimitar syndrome.” Pediatric Cardiac Surgery. Fourth Edition Ed. Constantine Mavroudis and Carl L Backer. Oxford, UK; Blackwell Publishing Ltd. 2013. 295-310.
5. Said SM, Burkhart HM, Schaff HV et. al. Single-patch, 2 patch, and caval division techniques for repair of partial anomalous pulmonary venous connections: Does it matter? J Thorac Cardiovasc Surg. 2012; 143: 896-903.
6. Sonavance SK, Milner DM, Aal AK, Shahir KS and Chautuvedi A. Comprehensive Imaging Review of the Superior Vena Cava. Radiographics. 2015 Nov-Dec; 35 (7): 1873-92.
Case prepared by: M. Jay Campbell, MD; Duke University







