Case Published in the Journal of Cardiovascular Magnetic Resonance: Click here for the link
Click here for PubMed Reference to Cite this Case
Clinical History
A 70-year-old female with a past medical history of asthma presented to the Emergency Department for further evaluation of atypical chest pain, orthopnea, and progressive dyspnea on exertion. Initial laboratory findings showed markedly elevated high-sensitivity troponin T (1,200 ng/L), NT-pro BNP (4,400 pg/mL), and hypereosinophilia (6.48 x 109/L). Her electrocardiogram (Image 1) demonstrated sinus tachycardia with diffuse ST depressions, new from prior studies.
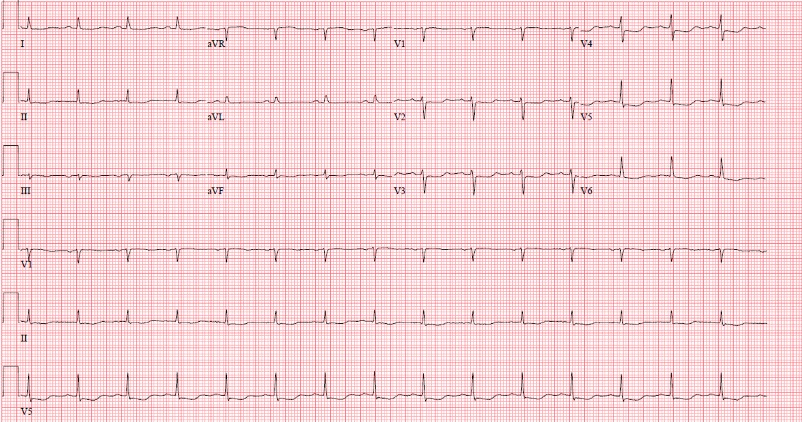
Image 1: A posteroanterior chest X-ray (Image 2) was notable for an enlarged cardiomediastinal silhouette with bilateral pleural effusions.

Image 2: Transthoracic echocardiography was notable for normal LV size, wall thickness, and systolic function. There was a moderate circumferential pericardial effusion and pericardial thickening (Video 1).

Video 1: A cardiac MRI (CMR) was ordered for further evaluation of suspected myopericarditis.
CMR Findings (3T)
Cine imaging demonstrated a moderate circumferential pericardial effusion and mild pericardial thickening (Video 2). LV cavity dimensions were small. Biventricular function was normal.
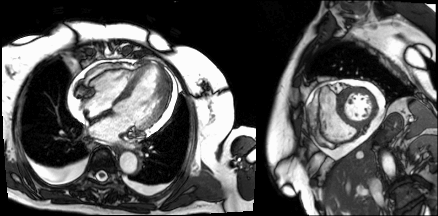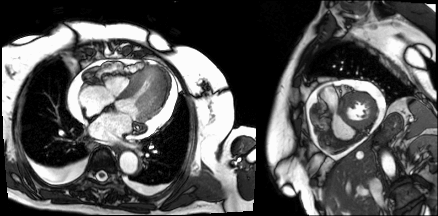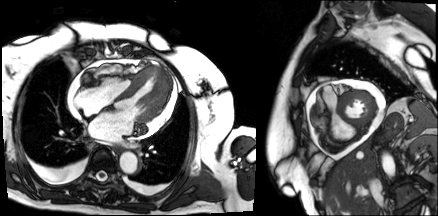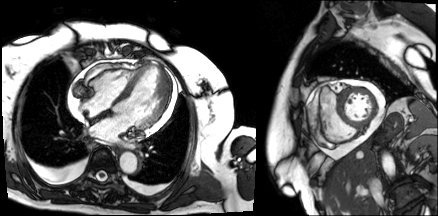
Video 1
The posterior right atrial wall briefly collapsed during atrial filling, suggesting increased pericardial pressures (Image 3). Importantly, this finding has relatively limited sensitivity (55-60%) and sensitivity (50-68%) for tamponade[1]. The specificity of right atrial collapse for tamponade improves when the collapse persists for more than 30% of the cardiac cycle, which was not observed in this case.
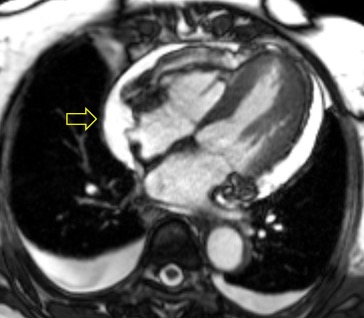
Image 3
Native T1 maps were deemed non-diagnostic due to poor co-registration from significant sinus arrhythmia, which led to image acquisition in different portions of the cardiac cycle. Automated motion correction, which adjusts for in-plane motion, may lead to errors in T1 measurements when both in-plane and through plane motion are present due to variations in the amount of partial volume between myocardium and blood. In this circumstance, manual correction leads to the same errors. T2 mapping did not reveal an elevation in T2, although only one slice was imaged.
Following administration of gadolinium contrast, single shot delayed enhancement imaging was performed using an inversion time of 875 ms (the null time for thrombus at 3T) to screen for intracardiac thrombus. Several hypo-enhanced (dark) regions were present along the endocardial surface of the left ventricle (Image 4), suggesting the presence of multiple LV thrombi (arrows).
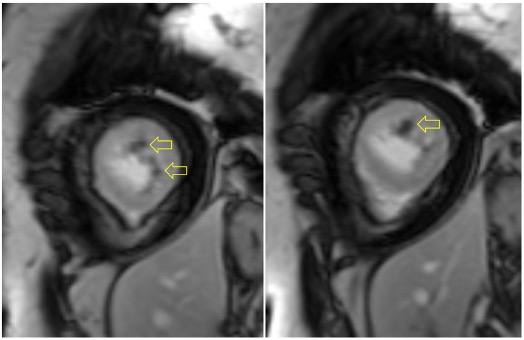
Image 4
Flow independent dark blood delayed enhancement (FIDDLE) imaging demonstrated regions of patchy, endocardial hyperenhancement involving the distal lateral wall, apex, anterior wall, and papillary muscles (arrows). These regions were not well visualized by conventional delayed enhancement imaging, likely due to the similar T1 of blood and the involved myocardium (Image 5). Pericardial enhancement was also noted.
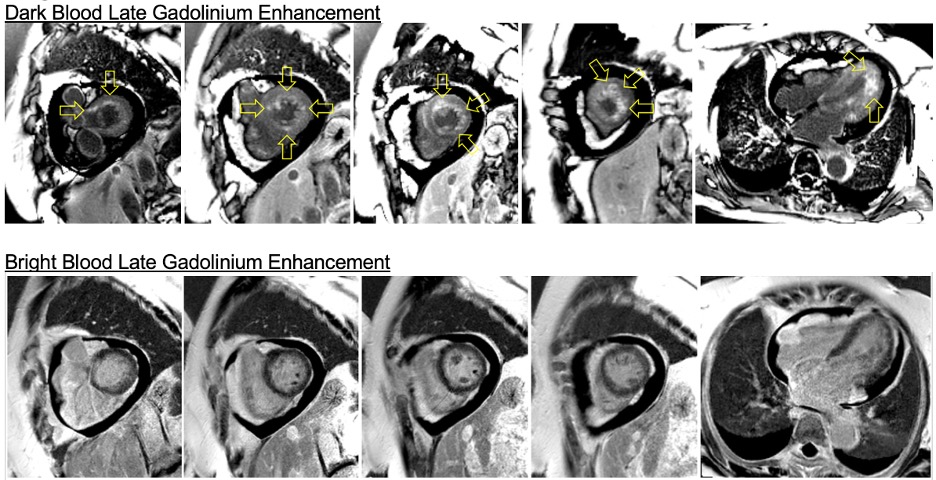
Image 5
Conclusion
This patient’s CMR findings (pericardial thickening and enhancement, LV thrombi, and patchy endocardial hyperenhancement of the distal segments) in the setting of new heart failure and severe hypereosinophilia are most consistent with acute eosinophilic myocarditis. A confirmatory endomyocardial biopsy was performed (Image 6).
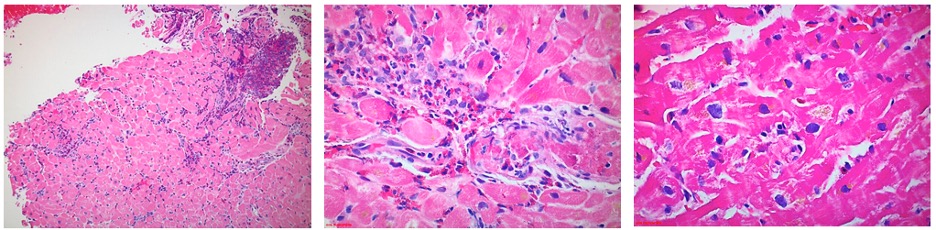
Image 6
The biopsy sample confirmed moderate perivascular and interstitial inflammatory cell infiltrate with prominent eosinophils. High dose steroid therapy was initiated with an extended prednisone taper. At the patient’s one-month post-discharge follow-up, she reported resolution of her dyspnea, orthopnea, and chest pain. Her laboratory abnormalities, ECG changes, and echocardiography findings normalized.
Perspective
Hypereosinophilic syndromes (HES) are a heterogenous group of conditions defined by end-organ damage due to the cytotoxic effects of eosinophilic infiltrates. The diagnosis is rare, with a reported prevalence of 0.036 cases per 100,000 [2]. Approximately half of all HES cases include cardiac involvement manifesting as eosinophilic myocarditis (EM) [3, 4]. EM progresses across three clinical stages: an acute necrotic stage, intermediate thromboembolic stage, and final fibrotic stage. This discussion will highlight the clinical features of each stage as well as the corresponding CMR findings.
The acute necrotic stage of EM, which is usually clinically silent, is characterized by markedly elevated troponin and NT-pro BNP levels in the setting of clean coronaries and normal ventricular function. Peripheral eosinophilia will remain absent in 25% of EM cases and cannot be used as a reliable indicator. CMR is highly sensitive for even the earliest stage of EM, characteristically showing hyperintensity on T1 or T2 weighted imaging with subendocardial late gadolinium enhancement [4, 5, 6].
Transition to the thromboembolic stage of EM is variable in timing and may occur 1 day to 3 months after the acute necrotic stage [7]. Symptom onset most often occurs during this second stage and might include fever (25-30% of cases), dyspnea (40-70%), atypical chest pain (40-45%), and thromboembolic complications (5%) [2, 8, 9, 10]. Intracavitary thrombogenesis in EM is driven by eosinophil peroxidase mediated production of hypothiocyanous acid, which enhances tissue factor expression and clot deposition in regions of dense eosinophilic infiltration [11]. This mechanism permits subendocardial thrombi to form despite the absence of wall motion abnormalities. Clot formation is easily visualized on CMR during this second stage through selective nulling of avascular tissue using prolonged inversion times [12].
The final fibrotic stage of EM is defined by scarring of the damaged myocardium, a restrictive cardiomyopathy, and new onset heart failure [13]. Preferential accumulation of thrombofibrotic material between the ventricular aspect of the posterior mitral leaflet and the mural endocardium of the left ventricular free wall may present as new mitral regurgitation [14]. Superimposed on the findings of the first and second stages, the presence of dilated atria (due to restrictive physiology and diastolic dysfunction) is a CMR finding that can be used to identify the fibrotic stage [15].
EM is a challenging diagnosis that demands a high clinical index of suspicion and a low threshold for acquisition of CMR. In our case, the diagnosis hinged upon the combination of localized hypertrophy evident on cine imaging and endocardial hyperenhancement on FIDDLE[16]. To our knowledge, this is the first case to highlight the use of FIDDLE in the diagnosis of EM, and demonstrates the importance of separating blood signal from hyperenhanced myocardium in patients with this disease.
At our center, we have used FIDDLE for clinical purposes since 2010. We have found that that utility of dark blood delayed enhancement extends beyond the detection of myocardial infarction and improves the visualization of hyperenhanced myocardium in a variety of patients with non-ischemic cardiomyopathies. Currently, FIDDLE is not commercially available, however, the works in progress sequence can be requested by participating members (with software compatibility) of the SCMR Registry.
Click here to review this case on Cloud CMR.
References
1. Restrepo, C.S., et al., Imaging findings in cardiac tamponade with emphasis on CT. Radiographics, 2007. 27(6): p. 1595-610.
2. Mubarik, A. and A.M. Iqbal, Loeffler Endocarditis. StatPearls, 2020.
3. Sen, T., et al., Cardiac involvement in hypereosinophilic syndrome. Tex Heart Inst J, 2009. 36(6): p. 628-9.
4. Bailey, G., et al., Eosinophilic Myocarditis Presenting as ST-segment Elevation Myocardial Infarction Diagnosed with Cardiac Magnetic Resonance Imaging. Am J Med, 2016. 129(2): p. e19-22.
5. Debl, K., et al., Time course of eosinophilic myocarditis visualized by CMR. J Cardiovasc Magn Reson, 2008. 10: p. 21.
6. Kim, E.Y., et al., Early non-invasive diagnosis and treatment of acute eosinophilic myopericarditis by cardiac magnetic resonance. J Korean Med Sci, 2011. 26(11): p. 1522-6.
7. Ogbogu, P.U., D.R. Rosing, and M.K. Horne, 3rd, Cardiovascular manifestations of hypereosinophilic syndromes. Immunol Allergy Clin North Am, 2007. 27(3): p. 457-75.
8. Sheikh, H., et al., The Clinicopathological Profile of Eosinophilic Myocarditis. Cureus, 2018. 10(12): p. e3677.
9. Dulohery, M.M., et al., Lung involvement in hypereosinophilic syndromes. Respir Med, 2011. 105(1): p. 114-21.
10. Brambatti, M., et al., Eosinophilic Myocarditis: Characteristics, Treatment, and Outcomes. J Am Coll Cardiol, 2017. 70(19): p. 2363-2375.
11. Wang, J.G., et al., The principal eosinophil peroxidase product, HOSCN, is a uniquely potent phagocyte oxidant inducer of endothelial cell tissue factor activity: a potential mechanism for thrombosis in eosinophilic inflammatory states. Blood, 2006. 107(2): p. 558-65.
12. Goyal, P. and J.W. Weinsaft, Cardiovascular magnetic resonance imaging for assessment of cardiac thrombus. Methodist Debakey Cardiovasc J, 2013. 9(3): p. 132-6.
13. Weller, P.F. and G.J. Bubley, The idiopathic hypereosinophilic syndrome. Blood, 1994. 83(10): p. 2759-79.
14. Gottdiener, J.S., et al., Two-dimensional echocardiographic assessment of the idiopathic hypereosinophilic syndrome. Anatomic basis of mitral regurgitation and peripheral embolization. Circulation, 1983. 67(3): p. 572-8.
15. Kharabish, A. and D. Haroun, Cardiac MRI findings of endomyocardial fibrosis (Loeffler’s endocarditis) in a patient with rheumatoid arthritis. J Saudi Heart Assoc, 2015. 27(2): p. 127-31.
16. Kim, H.W., et al., Flow-Independent Dark-blood DeLayed Enhancement (FIDDLE): validation of a novel black blood technique for the diagnosis of myocardial infarction. Journal of Cardiovascular Magnetic Resonance, 2016. 18(S1).
Case prepared by:
Sean G. Hughes, MD
Associate Editor
Vanderbilt University Medical Center







