Kristopher Pfirman DO1, Vishal Mehra MD PhD1, Shahzad Ahmed DO1
1Department of Cardiovascular Medicine, Geisinger Medical Center, Danville, PA
Clinical History
A 63-year-old male, with a known past medical history of left choroid melanoma presented with increasing dyspnea, hypertension, hyperlipidemia ongoing tobacco use, and progressive 30-pound weight loss and associated anorexia. Lab work was obtained indicating hyponatremia with a sodium of 128 mg/dL, elevated LDH of 293, and a leukocytosis of 21,300. A CT scan of the chest obtained 2 months prior to presentation identified numerous diffuse nodules throughout the lung fields with associated mediastinal and hilar lymphadenopathy (Figure 1). An echocardiogram showed a large sized echogenic mass noted in the left ventricle attached to the anterior septal wall measuring 1.9 by 1.6 cm (Figure 2). A second mass was located but difficult to completely visualize (Figure 3). A cardiovascular magnetic resonance (CMR) was requested to characterize the masses.
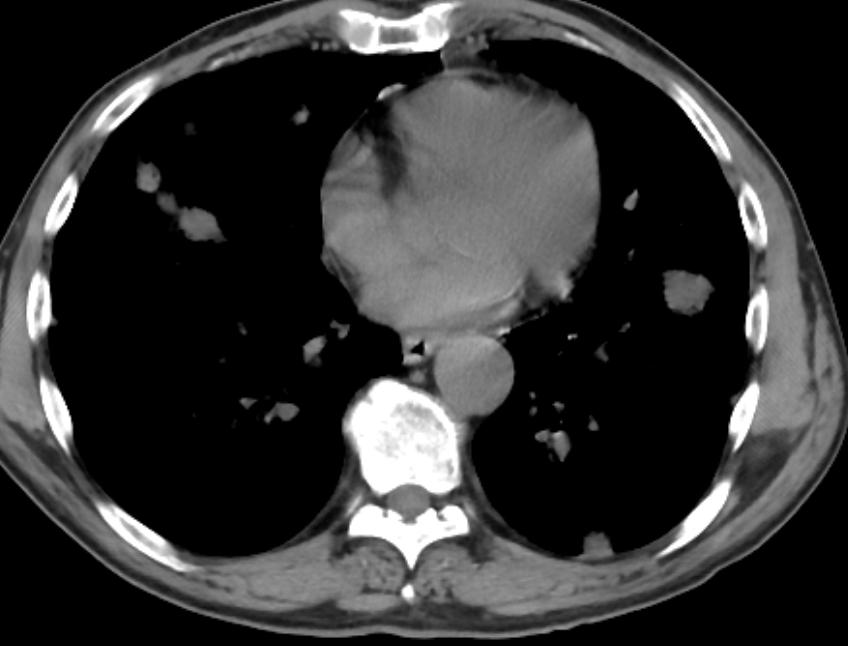
Figure 1. Computed tomography of the chest without contrast illustrating multiple lung lesions.
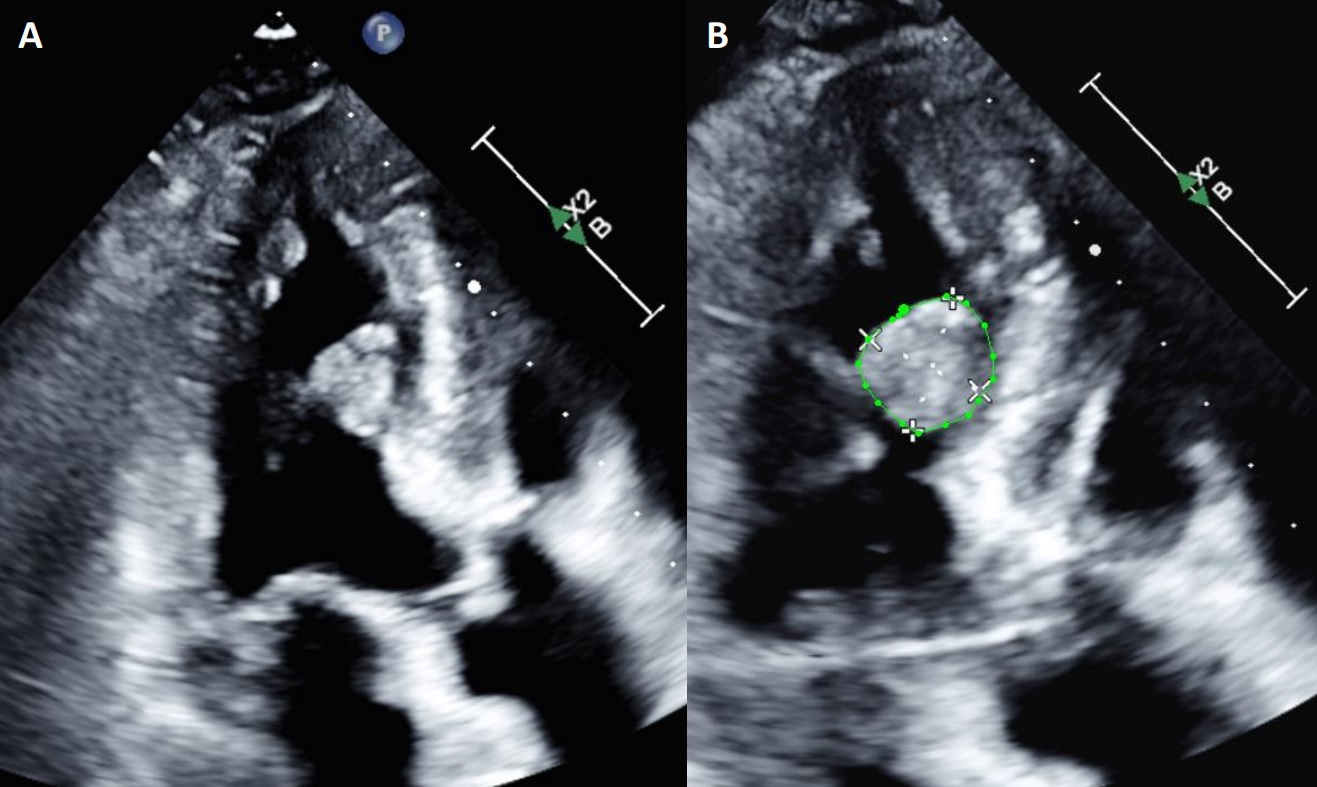
Figure 2. Transthoracic echocardiogram apical 3 chamber view without and with biplane measurements of a large myocardial mass.
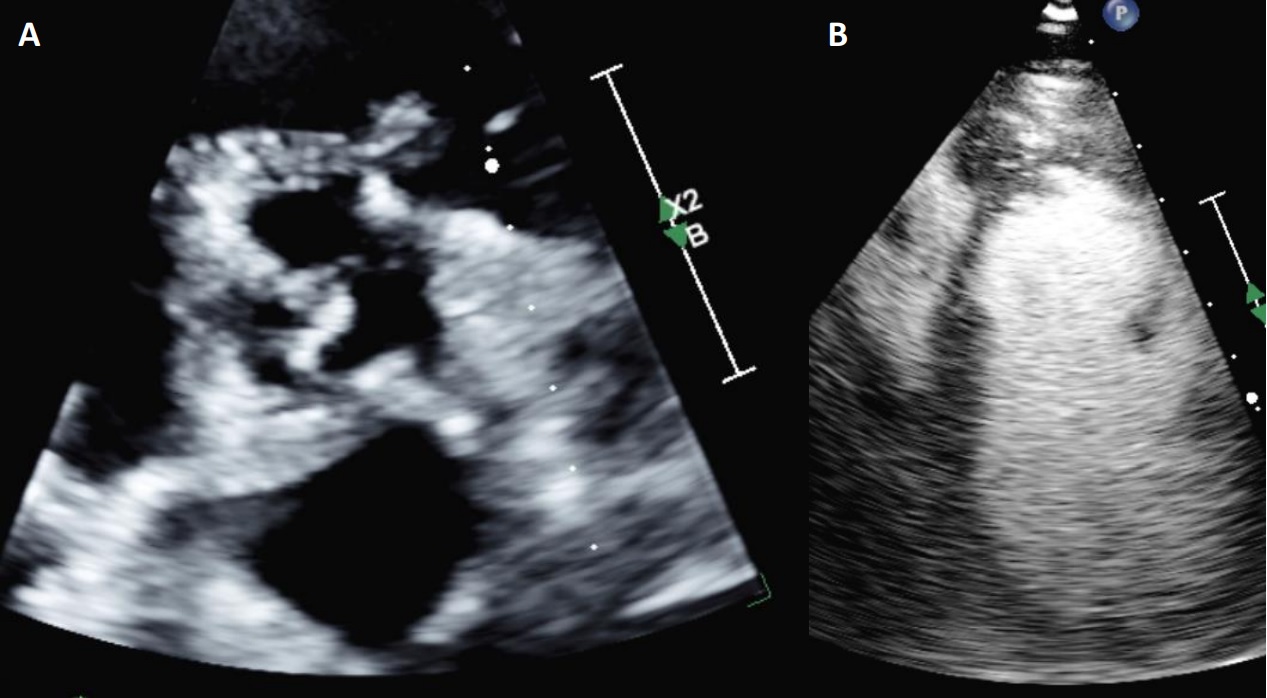
Figure 3. Transthoracic echocardiogram parasternal short axis view at the level of the aortic valve and a modified parasternal short axis with contrast enhancing agent to better identify the right ventricular/septal mass.
CMR findings
Multiple intramyocardial masses are noted in the inter-atrial septum, right atrium, left and right ventricles, largest measuring 16.5 x 13mm in the lateral LV wall (Movie 1). An anterior mass bulges into the LV cavity and an additional mass is protruding into the RV outflow tract from the anteroseptum. These heterogeneous, bulky, masses are hyperintense on cine SSFP as well as T2 and T1 black blood imaging (Figures 4-5). They are highly vascular on first pass rest perfusion (Movie 2) and have profound enhancement on late gadolinium imaging (Figure 6), extending into the subendocardial and subepicardial layers. This appearance, however, is likely due to the native T1 times being significantly shortened by the presence of melanin, rather than profound uptake of gadolinium within the tumors. This same phenomenon can also be seen with mitral annular calcification with caseous necrosis with short native T1 times. Similar masses are also noted along the pericardium, as well as innumerably disseminated throughout the lung fields that have the same tissue characterization as the myocardial metastases. Mediastinal and hilar lymphadenopathy are also present. A low native T1 time ~700ms was observed (Figure 7). A moderate sized (5-10 mm) circumferential pericardial effusion is present. Real-time, free breathing sequences were utilized when possible due to the patient’s dyspnea and inability to adequately perform breath holds.

Movie 1. Cine SSFP short axis stack showing multiple intramyocardial masses in the left and right ventricles.

Figure 4. T1-weighted SPAIR images. Spin Echo sequence. Left = mid-short axis view. Right = 4 chamber view.

Figure 5. T2-weighted SPAIR images. Spin Echo sequence. Left = mid-short axis view. Right = 4 chamber view.
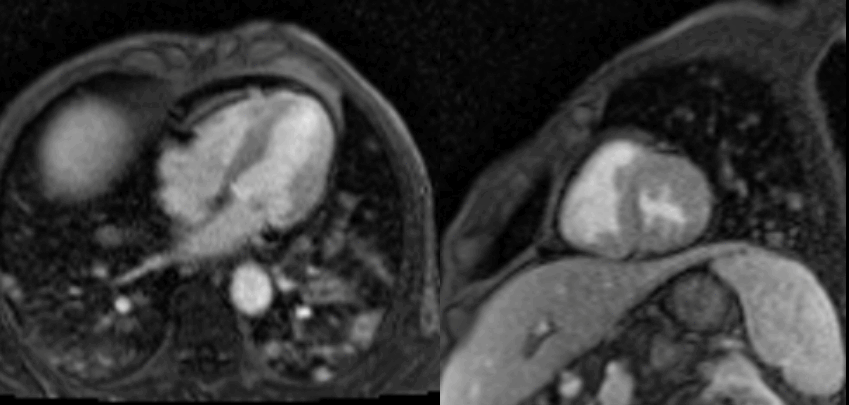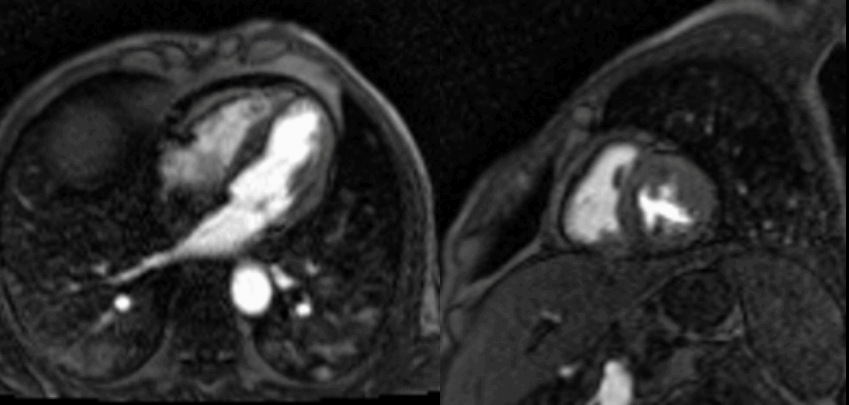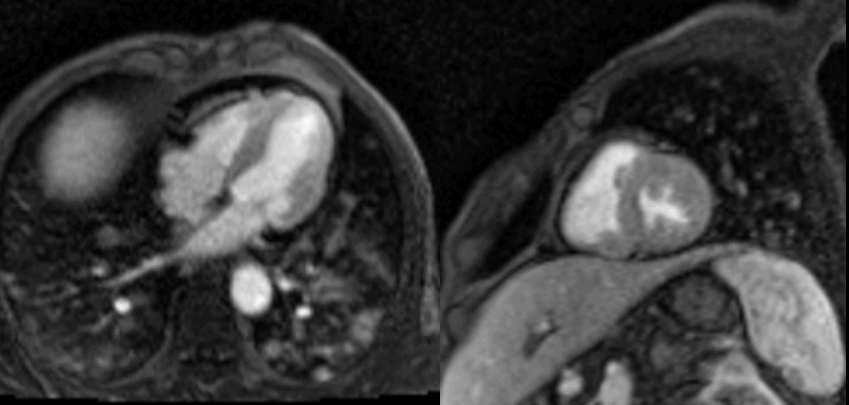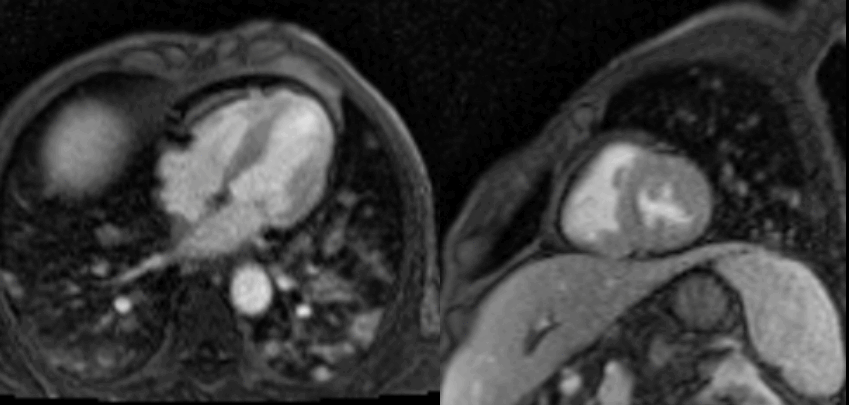
Movie 2. Four chamber and mid short axis rest perfusion imaging showing brisk uptake of contrast of first pass perfusion.
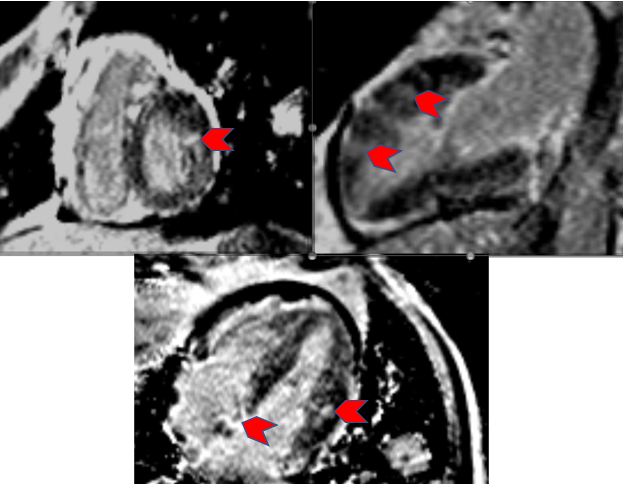
Figure 6. Delayed gadolinium imaging sequences. Gradient Recovery/Inversion Recovery sequences. PSIR images. Top left = Basal short axis view. Top right = 2 chamber/vertical long axis view. Bottom = 4 chamber/horizontal long axis view. Chevrons = Late gadolinium enhancement of metastasis.

Figure 7. Native T1 and T2 parametric mapping images from the mid short axis view. Top image: T1 parametric mapping. Arrow indicates low T1 times (metastasis). Bottom image: T2 parametric mapping. Arrow = 52 msec. Chevron = 75 msec (elevated).
Conclusion
CMR findings were consistent with an underlying malignant process suggestive of a metastatic disease from known prior primary left choroid melanoma. The presence of highly-melanin rich metastatic tumors affects the T1 relaxation times. The T1 relaxation times shorten and recover their signal faster than the surrounding normal myocardium that results in high signal on the T1-weighted fast spin echo sequences. This is a quintessential finding that helps differentiate malignant melanoma from other metastatic cardiac tumors. The T2-weighted images also in this case exhibited high signal which may or may not be elevated and is also dependent on the amount of surrounding edema. The T1 shortening effects of melanin dominates over the T1 prolongating of myocardial inflammation and edema. Malignant melanoma is unique in that it is one of the times where native T1 and T2 times are discordant despite the evidence of edema and necrosis. Given the highly vascularized nature of these tumors, the first pass perfusion is usually preserved, and appears as bright as normal myocardium due to high vascularity. A pericardial effusion is also present in most cases, traditionally exhibits hemorrhagic features. These complex features of the fluid consist of intermediate T2-weighted signal intensity and high T1-weighted signal intensity. The SSFP cine images may identify loculations including fibrin strands and thrombi from the dissemination and seeding of the pericardium.
A suspicion of malignant melanoma was raised from the initial work-up given the findings on the chest CT scan and transthoracic echocardiogram. Given persistence of the patient’s symptoms in the setting of concerning findings on CMR suggesting disseminated malignant melanoma, he was admitted to the hospital for further management by hematology/oncology to initiate immunotherapy.
Based on CMR findings, a full body FDG-PET/CT imaging was performed confirming widespread metastatic disease including the central nervous system. The diagnosis of metastatic malignant melanoma was made and further management with nivolumab, ipilimumab, and allopurinol was implemented. The patient was discharged from the hospital and has elected to continue treatment as an outpatient.
Perspective
Tissue characterization is an extremely beneficial when identifying the etiology of myocardial masses. The dimensions, morphology, location and any associated hemodynamic or mass effect, mobility, vascularity, infiltration patterns, magnetic properties, and the presence of associated sequelae such as effusions can help with differentiating the underlying etiology of cardiac masses. The size of benign tumors rarely surpasses 5 cm (1). Malignant tumors are more likely to infiltrate into the adjacent structures, extend with irregular borders, and demonstrate associated pericardial effusions (2). The native magnetic properties of tumors as well as the effects of late gadolinium enhancement can aid in non-invasive tissue characterization. All malignant tumors are likely to exhibit late gadolinium enhancement and first pass resting perfusion imaging due to their vascularity can aid in differentiating malignancy from cardiac thrombi. Prolonged inversion times of greater than or equal to 422 msec has been shown to aid in the delineation of cardiac thrombi from masses on late gadolinium imaging sequences (1). When all of the above is taken into account, diagnostic accuracy has been as high as 92 to nearly 100% in prior studies published (2).
Metastatic melanoma represents approximately 27.8% of metastatic malignancies involving the heart; most commonly the right atrium. This equates to an incidence of about 1-5% (1). CMR has a pivotal role in the differential diagnosis of melanoma from the other disseminated malignancies given the melanin-rich characteristics of the tumors. These tumors are thought to be spread by hematogenous dissemination in most cases (3). This results in multiple organ system involvement as seen in our case with the central nervous, pulmonary, lymphatic, and cardiac systems inundated with tumor burden.
The unusual pattern of melanoma and significantly reduced T1 relaxation times resulting in enhanced signal intensity is a hallmark feature differentiating it from other cardiac masses. Although rare the survival rates are dismal with the 5-year survival rate of 15-20% with the rate of mortality falling from of all malignant melanomas by 5.7%, annually (4). The overall mean survival rate for stage 4 malignant melanoma was 9.2 month with a median survival of 6.3 months (5). CMR aids in the diagnosis of malignant melanoma, and relevant implications on patients’ management, but unfortunately given the increased mortality risk associated with this disease, our patient’s overall prognosis is poor.
Click here to view the entire study on CloudCMR.
References:
1. Lombardi M. “Section 8, Chapter 5 – Metastatic Tumours.” The EACVI Textbook of Cardiovascular Magnetic Resonance, Oxford University Press, Oxford, 2018, pp. 463–483.
2. Kassi M, et al. Differentiating benign from malignant cardiac tumors with cardiac magnetic resonance imaging. J Thorac Cardiovasc Surg. May 2019;157(5):1912-1922.
3. Villa A, et al. Cardiac metastases of melanoma as first manifestation of the disease. J Radiol Case Rep. 2014;8(4):8-15.
4. Siegel RL, et al. Cancer Statistics, 2021. CA Cancer J Clin. 2021;71(1):7-33.
5. Sandru A, et al. Survival Rates of Patients with Metastatic Malignant Melanoma. J Med Life. 2014;7(4):572-576.
Case prepared by:
Jason N. Johnson, MD MHS
Associate Editor, SCMR Case of the Week
Le Bonheur Children’s Hospital, University of Tennessee Health Science Center, St. Jude Children’s Research Hospital







