Madhusudan Ganigara, MD, Mohamed Nagiub, MD, Tarique Hussain, MD, PhD
Division of Pediatric Cardiology, Children’s Medical Center Dallas, Dallas, Texas, USA
Clinical History:
A near term newborn with maternal history of chorioamnionitis was admitted to the neonatal ICU for respiratory distress after birth. He was managed as per the unit protocol for neonatal sepsis. He was soon noted to have persistent atrial tachycardia requiring rate control with medical therapy, after attempts at cardioversion with vagal maneuvers, adenosine and synchronized cardioversion failed. Initial transthoracic echocardiogram revealed normal cardiac anatomy with normal myocardial contractile function. He subsequently developed myocardial dysfunction and low cardiac output state requiring inotropic support. There was mild troponin leak and detection of enterovirus in blood by PCR raising suspicion of neonatal myocarditis.
A cardiac MRI at 2 weeks of age revealed depressed left ventricular contractile function (ejection fraction 41%) with no late gadolinium enhancement (LGE). (Image 1)
Patient clinically improved with supportive therapy. Follow up serial echocardiograms revealed improving LV systolic function with new findings of patchy subendocardial echo-brightness in the basal- and mid-septal walls. A repeat cardiac MRI was requested at 4 weeks of age.

Image 1: Late gadolinium enhancement (LGE) imaging showing no delayed myocardial enhancement at the 2-week scan.
CMR Findings:
The repeat cardiac MRI using neonatal protocol was performed using Phillips Ingenia 1.5 T scanner. This included avoiding sedation using ‘feed and swaddle’ method [1]. This revealed normal left ventricular dimensions with normal ventricular mass and systolic function (measured ejection fraction 71%). There was diminished systolic thickening of the basal anteroseptal and basal inferoseptal LV segments (AHA segments 2,3). (Video 1)
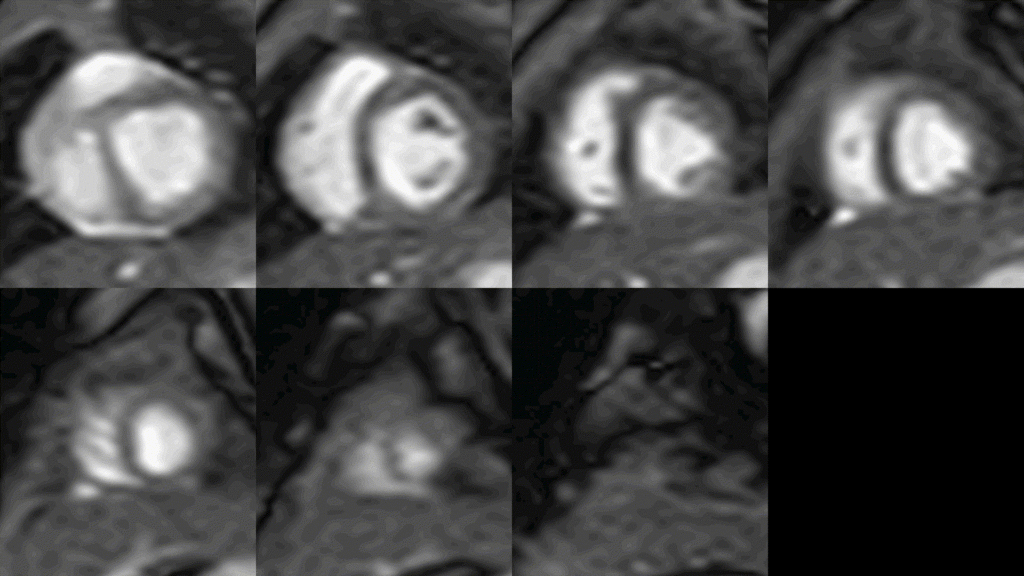
Video 1: Short-axis SSFP cine demonstrating normal left and right ventricular size and global systolic function. There was diminished systolic thickening of the basal anteroseptal and basal inferoseptal LV segments (AHA segments 2 and 3).
LGE imaging was performed immediately after administration of Gadolinum contrast [Gadavist® (Bayer HealthCare Pharmaceuticals)] to account for the rapid washout of contrast in the setting of high neonatal heart rates. A respiratory motion-compensated free-breathing gradient echo T1-weighted phase sensitive inversion recovery (PSIR) sequence was used. Inversion time was selected from a modified Look-Locker sequence based on the inversion time where myocardium was best nulled. Thin (6 mm) myocardial slices in the short axis were acquired through the ventricles with a field of view of 200 mm and inplane resolution of 1.6 x 1.9 mm.
Acceleration techniques included a SENSE factor of 1.8 and signal averaging (NSA) of 2. Mid diastolic imaging with shortened read-out window (50ms) was obtained at every 4th R-R interval to account for the fast heart rate. This revealed sub-endocardial partial thickness delayed myocardial enhancement of the AHA segments 2 and 3. (Image 2) These findings were consistent with neonatal thromboembolic myocardial infarction.
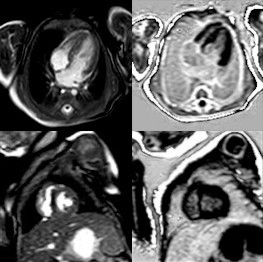
Image 2: Sub-endocardial partial thickness delayed myocardial enhancement of the AHA segments 2 and 3 (Above panel 4CV and lower panel mid-LV SA).
Conclusion:
Neonatal myocardial infarction in the infant is suspected to be the result of a perinatal thromboembolic event. Paradoxical embolism through the foramen ovale or the ductus arteriosus can occur in the fetus in association with umbilical cord hematoma, umbilical venous catheterization, prenatal renal vein thrombosis and many times, without obvious sources [2].
The patient continued to improve clinically with supportive therapy and was discharged at 4 weeks of age. Since then, he has been asymptomatic, showing normal growth and development, no new arrythmias and normal cardiac function. He was continued on betablocker therapy for arrythmia control with no significant side effects.
Perspective:
Late gadolinium enhancement (LGE) cardiac magnetic resonance (MR) imaging sequence is increasingly used in the evaluation of pediatric cardiovascular disorders. It is seen as a result of extracellular space expansion, either from acute cell damage or chronic scarring or fibrosis. LGE imaging in neonates and young infants is challenging but feasible.
The challenge of imaging in the setting of high neonatal heart rates is overcome using acceleration techniques and adjusting the acquisition window (views per segment) to the heart rate [2, 3]. To account for the small heart size in neonates using a smaller field of view (200 mm) and thinner slices (5-6 mm) gives an adequate spatial resolution. These measures, however, decrease the voxel size resulting in a low signal-to-noise (SNR) ratio. This is improved by using the smallest available coil (eg. a smaller pediatric multi-element cardiac phased-array coil) and signal averaging. Motion compensation is used to account for the respiratory motion.
Typically, 180-degree inversions are placed every two R-R intervals for LGE sequences to allow for recovery of the longitudinal signal between successive inversion pulses. However, this is not feasible at high heart rates and triggering is altered to every 4th R-R interval in neonates. This may require manually halving the heart rate to ‘trick’ the scanner into increasing the acquisition window. Lastly, neonates have a higher rate of wash out of the Gadolinium contrast agent from the myocardium, necessitating minimal delay in obtaining the LGE images after contrast administration.
Click here to view the entire case on CloudCMR.
References:
Case prepared by:
Madhusudan Ganigara, MD
Associate Editor, Cases of SCMR; UT Southwestern Children’s Medical Center







