Salomon Chamay MD, Stephen Horgan MD, Seth Uretsky MD
Division of Cardiology, Morristown Medical Center, Morristown, NJ
Clinical History
We present the case of a 66-year-old Caucasian female with history of celiac disease, hypothyroidism, and ovarian cancer who presented with progressively worsening dyspnea of two weeks duration. Initial vital signs were unremarkable with a presenting blood pressure of 136/69 mmHg. Physical examination was notable for decreased breath sounds over bilateral bases. ECG showed normal sinus rhythm with left atrial enlargement and laboratory studies were remarkable for an elevated NT-proBNP of 2,718 pg/mL (Reference <450 pg/mL). Computed tomography angiography of the chest revealed bilateral pleural effusions and no evidence of pulmonary embolism. A transthoracic echocardiogram (TTE) was performed which raised suspicion for moderate to severe eccentric tricuspid regurgitation (TR) with a velocity of 4-4.5 m/s, estimated right ventricular pressure of 70 mmHg, and normal left ventricular ejection fraction (Movie 1). Cardiac magnetic resonance (CMR) imaging was performed to further evaluate the eccentric TR and new-onset heart failure.
 |
| Movie 1. Color compare Doppler TTE subcostal coronal view with suspected moderate to severe TR and normal bi-ventricular systolic function. |
CMR Findings
A CMR study was performed using a 1.5T scanner (Artist, General Electric, Milwaukee Wisconsin, USA). Short axis and aortic valve balanced steady state free precession (bSSFP) stacks along with orthogonal long axis imaging demonstrated rupture of the noncoronary sinus of Valsalva with left to right shunting of blood into the right atrium (Movies 2-4, Figures 1-3). No aneurysm was noted within the sinus or aortic root. A jet from the fistula was noted to impinge on the septal leaflet of the tricuspid valve. Analysis of TR was limited by the presence of the significant shunt, however, the initial TTE finding of severe TR was attributed to the fistula. Phase contrast imaging of the pulmonary artery and ascending aorta revealed a significant left-to-right shunting with a Qp:Qs of 3.2:1 (Figure 4). The aortic root measured 4.1 cm in diameter, the aortic valve was trileaflet, and “aortic insufficiency” or volume of blood through the ruptured sinus into the right atrium was quantified at a volume of 26 mL and “regurgitant fraction” of 47% (Figure 5). Biatrial enlargement was noted, and the measured left ventricle end-diastole indexed (LVEDVI) and right ventricle end-diastole indexed (RVEDVI) were 88 mL/m2 and 108 mL/m2, respectively.
 |  |
| Movie 2 and Figure 1. Basal short axis cine bSSFP with a ruptured noncoronary sinus of Valsalva aneurysm (yellow arrow) with left to right shunting into the right atrium. | |
 |  |
| Movie 3 and Figure 2. Aortic valve cine bSSFP with a ruptured noncoronary sinus of Valsalva aneurysm (yellow arrow) with left to right shunting into the right atrium. | |
 |  |
| Movie 4. Off axis left ventricular outflow tract cine bSSFP with a ruptured noncoronary sinus of Valsalva aneurysm (yellow arrow) with left to right shunting into the right atrium. | |
 | |
| Figure 4. Phase contrast flow analysis of main pulmonary artery and ascending aorta with a calculated Qp:Qs of 3.2:1. | |
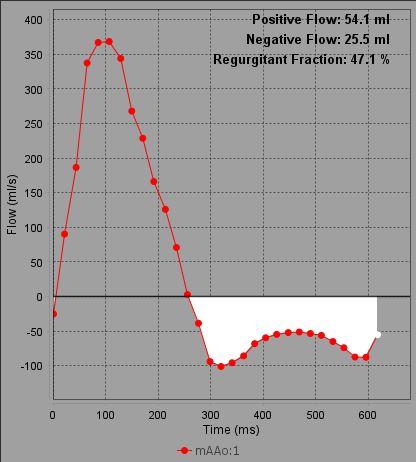 | |
| Figure 5. Quantified aortic regurgitation on flow analysis. | |
Conclusion
A diagnosis of acutely ruptured noncoronary sinus of Valsalva with fistulous flow into the right atrium was made. This was further confirmed on transesophageal echocardiography (TEE) (Movie 5) and coronary computed tomography angiography (CorCTA) (Figure 6). Cardiothoracic surgery was consulted due to concerns that the patient’s symptoms were secondary to the fistula. Intraoperatively, a large fistula originating at the noncoronary cusp draining into the RA was noted. The patient underwent successful repair with a bovine pericardial patch. A portion of the cusp was removed intraoperatively and sent to pathology, which reported myxoid fibroconnective tissue with degenerative changes and no signs of acute infection. The patient’s symptoms markedly improved, and she was seen at 6 weeks and 4 months postoperatively with complete resolution of her symptoms. A repeat CMR performed 1 year postoperatively demonstrated a normal Qp:Qs with no residual shunting (Movie 6). On repeat CMR, the right ventricular volume had decreased with an RVEDVI of 86 mL/m2.
 |
| Movie 5. TEE color Doppler at 45 degree view showing a ruptured sinus of Valsalva with shunting into the right atrium. |
 |
| Figure 6. Coronal CorCTA showing a ruptured sinus of Valsalva aneurysm from the non-coronary cusp to the right atrium. |
 |
| Movie 6. Aortic valve cine bSSFP with normal valve mobility and repair of the sinus of Valsalva rupture with no shunting from the aorta to the right atrium. |
Perspective
In this case, CMR led to an accurate diagnosis and identification of the etiology of the patient’s new onset heart failure. Sinus of Valsalva rupture is a rare aortic root pathology, most often associated with sinus aneurysms. Rarely, aneurysmal dilation and rupture may occur secondary to congenital anatomy, infection such as endocarditis, trauma or iatrogenic, or connective tissue disease. Sinus of Valsalva aneurysms have a prevalence of 0.09% in the general population based on autopsy data, with rupture occurring in a third of cases. [1] Sinus of Valsalva aneurysms are more common in Asian populations, are discovered in the third and fourth decades of life, and affect men more frequently than women with a 4:1 ratio. [2,3] While most unruptured sinus of Valsalva aneurysm cases are asymptomatic, rupture of the sinus often leads to symptoms including dyspnea, chest pain, insidiously progressive congestive heart failure, and rarely cardiac arrest. [4] Ruptured sinus of Valsalva aneurysms involve the right coronary cusp most frequently (61%), followed by the non-coronary cusp (26%), and left coronary cusp (12%). [5,6] Approximately 39% of cases rupture into the right atrium (21% from the non-coronary cusp, 17% from the right coronary cusp), while 31% of ruptured right coronary cusps flow into the right ventricular outflow tract. [5] Rupture without the presence of an aneurysm, such as in our case, has rarely been described and has been linked to minimal aortic root dilation or an occult aneurysm.
Most cases of sinus of Valsalva rupture are diagnosed with either TTE or TEE, although cardiac computed tomography has recently gained increased popularity. [2] CMR is often used as supplemental or confirmatory testing and allows for evaluation of intracardiac shunts in cases of rupture. Of 177 case reports of sinus of Valsalva aneurysms, 19 cases (11%) described CMR imaging, which were all performed to further define cardiac anatomy in known or suspected sinus of Valsalva aneurysms. [4] Advantages of CMR include avoidance of ionizing radiation, quantification of intracardiac shunting, identification of aortic regurgitation, ability to evaluate left ventricular hemodynamics and flow, and accurate assessment of the size and origin of the aneurysm. [7]
Our patient did not represent a classic example of sinus of Valsalva rupture in that she was female, in her sixth decade of life, Caucasian, and did not have clear aneurysmal dilation of the sinus or aortic root. Furthermore, the ruptured sinus was not identified on initial TTE and instead was discovered and diagnosed on CMR imaging. In this case, CMR led to an accurate diagnosis, localization of the fistula, and quantification of the degree of shunting, allowing for prompt surgical planning and treatment.
Click here to review the CMR on CloudCMR.
References
- Iwakawa H, Iino T, Yamamoto H, Watanabe H. Large non-coronary sinus of Valsalva aneurysm: a case report of an unusual cause of angina pectoris. Eur Heart J Case Rep. 2020 Sep 7;4(5):1-6.
- Weinreich M, Yu PJ, Trost B. Sinus of valsalva aneurysms: review of the literature and an update on management. Clin Cardiol. 2015 Mar;38(3):185-9.
- Miranda D, Peter AA, Osorion J, Ferreira AC. Ruptured aneurysm of the noncoronary sinus of valsalva. Tex Heart Inst J. 2005;32(4):586-8.
- Bricker AO, Avutu B, Mohammed TL, Williamson EE, Syed IS, Julsrud PR, Schoenhagen P, Kirsch J. Valsalva sinus aneurysms: findings at CT and MR imaging. Radiographics. 2010 Jan;30(1):99-110.
- Marroush TS, Boshara AR, Botros B, Vendittelli P, Ahmed Z, Dawood L, Rosman HS. Rupture of sinus of Valsalva aneurysm: Two case reports and a concise review of the literature. Heart Lung. 2018 Mar-Apr;47(2):131-135.
- Dayco JS, Dsouza A, Baciewicz F, Cardozo S. A rare variant of a ruptured sinus of valsalva aneurysm forming an aorto-atrial fistula: a case report and review of literature. Eur Heart J Case Rep. 2023 Feb 1;7(2):ytad018.
- Xu B, Kocyigit D, Betancor J, Tan C, Rodriguez ER, Schoenhagen P, Flamm SD, Rodriguez LL, Svensson LG, Griffin BP. Sinus of Valsalva Aneurysms: A State-of-the-Art Imaging Review. J Am Soc Echocardiogr. 2020 Mar;33(3):295-312.
Case prepared by:
Jason N. Johnson, MD MHS
Editor-in-Chief, Cases of SCMR
Le Bonheur Children’s Hospital, The University of Tennessee Health Science Center, St. Jude Children’s Research Hospital







