Case from: Srinivas Narayan, Martin Grips, Tarique Hussain
Institute: Evelina London Children’s Hospital.
Clinical history:
A 3-year-old girl was referred for evaluation of breathlessness on exertion (NYHA class 3) in conjunction with cyanosis. She was born at term without any perinatal problems and was growing and developing normally. Her parents noticed her becoming very breathless and blue around her mouth during exertion about 5-6 months before presentation. She did not have any other symptoms such as chest pain, palpitations, or loss of consciousness and there was no family history of any congenital heart disease.
On examination she was centrally cyanosed with a saturation of 88% in air. She had a hyperactive precordium with a right ventricular heave and palpable P2. There was a soft 1-2/6 systolic murmur over the left lower sternal border.
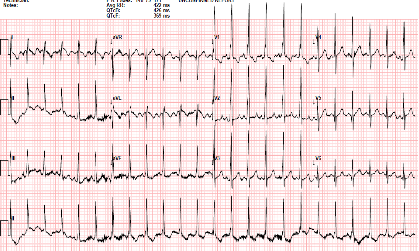
Figure 1: 12-lead EKG showing right-axis deviation and RVH
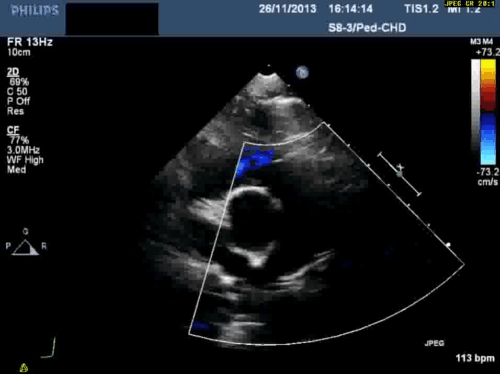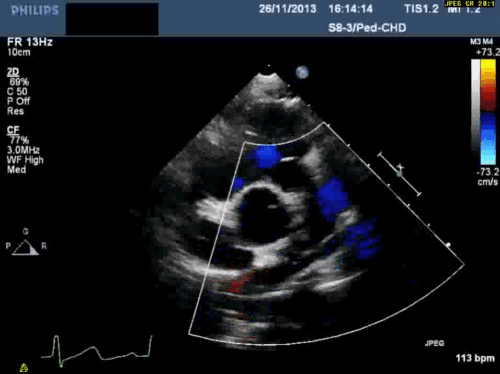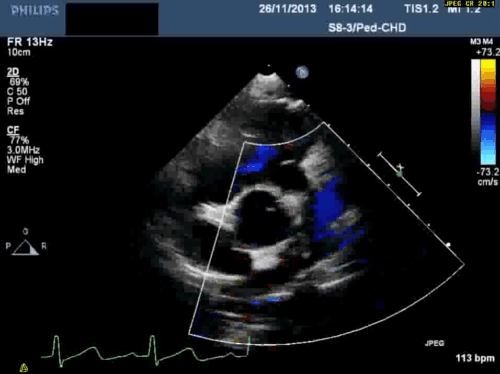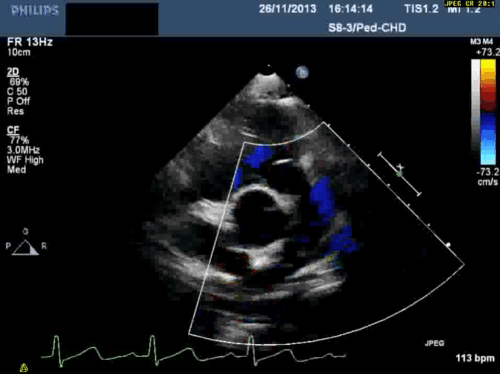
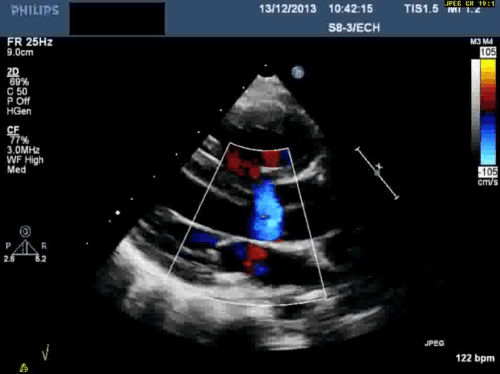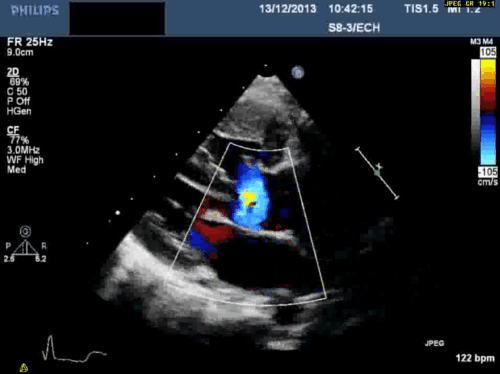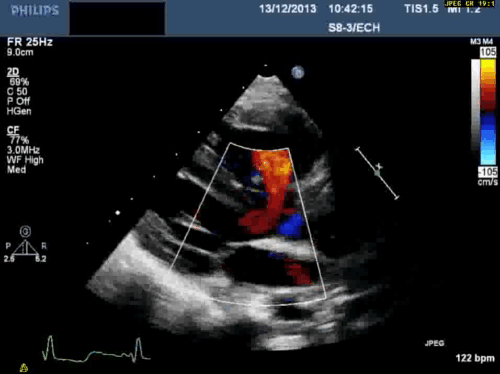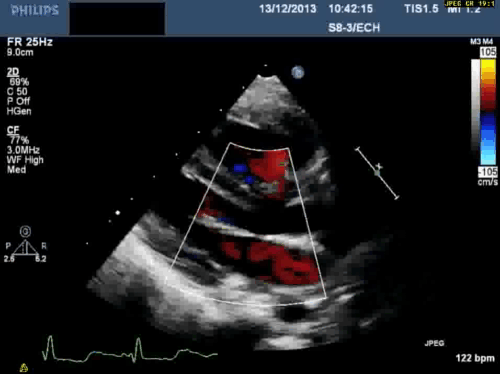
In addition, echocardiography showed that the aortic arch was right-sided. Pulmonary venous drainage appeared normal.
The clinical team wished to evaluate the cardiac anatomy, in particular the origin of LPA and whether there was a fixed and increased pulmonary vascular resistance. A comprehensive XMR (cardiac catheter & MRI in a dedicated combined hybrid lab) protocol was therefore performed.
CMR Findings:
The procedure began with a high temporal resolution angiogram (also known as 4D-TRAK (Time-resolved angiography using Keyhole)).

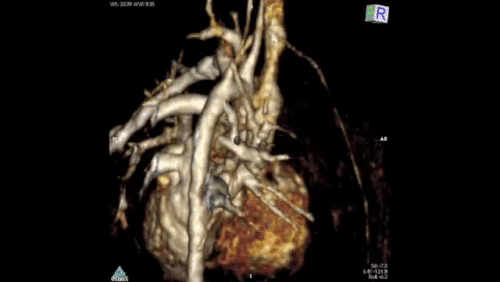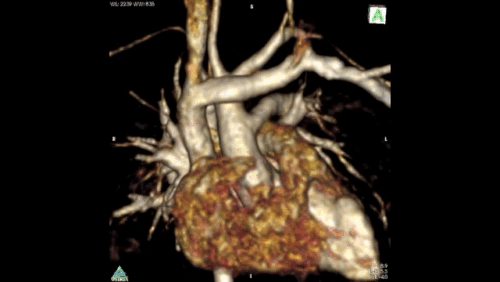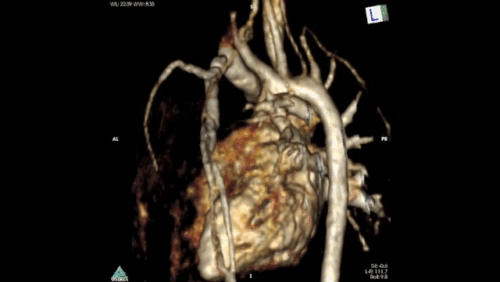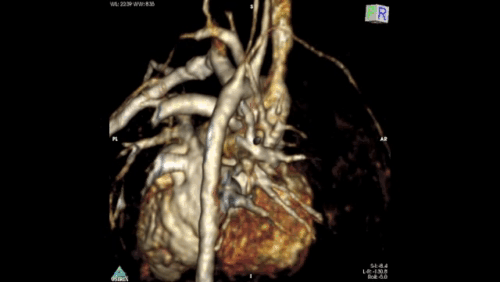
Figure 2: Angiography immediately demonstrated that the child has a Hemitruncus with the LPA arising from the ascending aorta. This is shown using volume rendering above. A right arch with normal branching is also seen.
Once the anatomy was known, the patient was immediately trasferred to the X-ray angiogrphy end of the room to undergo diagnostic cardiac catheter. Non-ferromagnetic catheters were placed anterograde in the RPA and retrograde in the LPA. The patient was transferred back to MRI for haemodynamic measurements including balanced SSFP cine images and selected phase contrast flow images. This was done under two conditions –
Condition 1. Baseline: 30% inhaled oxygen and
Condition 2: 100% inhaled oxygen and inhaled Nitric Oxide at 20 parts per million
The clinical importance of the XMR facility was demonstrated in this case. Time-resolved angiography was rapidly able to give the anatomical diagnosis. The attending physicians could then progress to accurately determining pulmonary vascular resistance, in order to assess for operability. This type of split lung anatomy and vascular resistance calculation is only possible using the XMR facility. It would not be feasible or meaningful using traditional catheterisation with application of the Fick principle. (ref 4)
The resistance calculation demonstrated the the condition was not operable and symptomatic care was pursued.
Perspective:
XMR facility allows a rapid diagnostic service with invaluable clinical information. This type of accurate diagnostic information is not possible using traditional means and higlights the necessity for all units to have access to XMR facilities for complex congenital heart disease cases.
References:
- Razavi R, Hill DL, Keevil SF, Miquel ME, Muthurangu V, Hegde S, Rhode K, Barnett M, van Vaals J, Hawkes DJ, Baker E. Cardiac catheterisation guided by mri in children and adults with congenital heart disease. Lancet. 2003;362:1877-1882
- Kuehne T, Yilmaz S, Schulze-Neick I, Wellnhofer E, Ewert P, Nagel E, Lange P. Magnetic resonance imaging guided catheterisation for assessment of pulmonary vascular resistance: In vivo validation and clinical application in patients with pulmonary hypertension. Heart. 2005;91:1064-1069
- Bell A, Beerbaum P, Greil G, Hegde S, Toschke AM, Schaeffter T, Razavi R. Noninvasive assessment of pulmonary artery flow and resistance by cardiac magnetic resonance in congenital heart diseases with unrestricted left-to-right shunt. JACC. Cardiovascular imaging. 2009;2:1285-1291
- Talwar S, Meena A, Ramakrishnan S, Choudhary S, and Airan B. Hemitruncus with ventricular septal defect in a 6-year-old child. Ann Pediatr Cardiol. 2013 Jul-Dec; 6(2): 194–196.
COTW handling editor: Tarique Hussain, MD





