Shaaban M1,2, Naguib A1, Haroun D1, Elkafrawy F1, Khalil S1, Tantawy S1, Elmozy W1, Romeih S1,2
1 Aswan Heart Centre, Aswan, Egypt.
2 Cardiology department, Faculty of Medicine, Tanta University, Egypt.
Clinical history
A 32-year-old male patient with no relevant medical history presented with acute onset of chest pain. His initial ECG showed ST segment elevation in leads V1 to V3 with reciprocal ST segment depression in inferior leads (Figure 1).
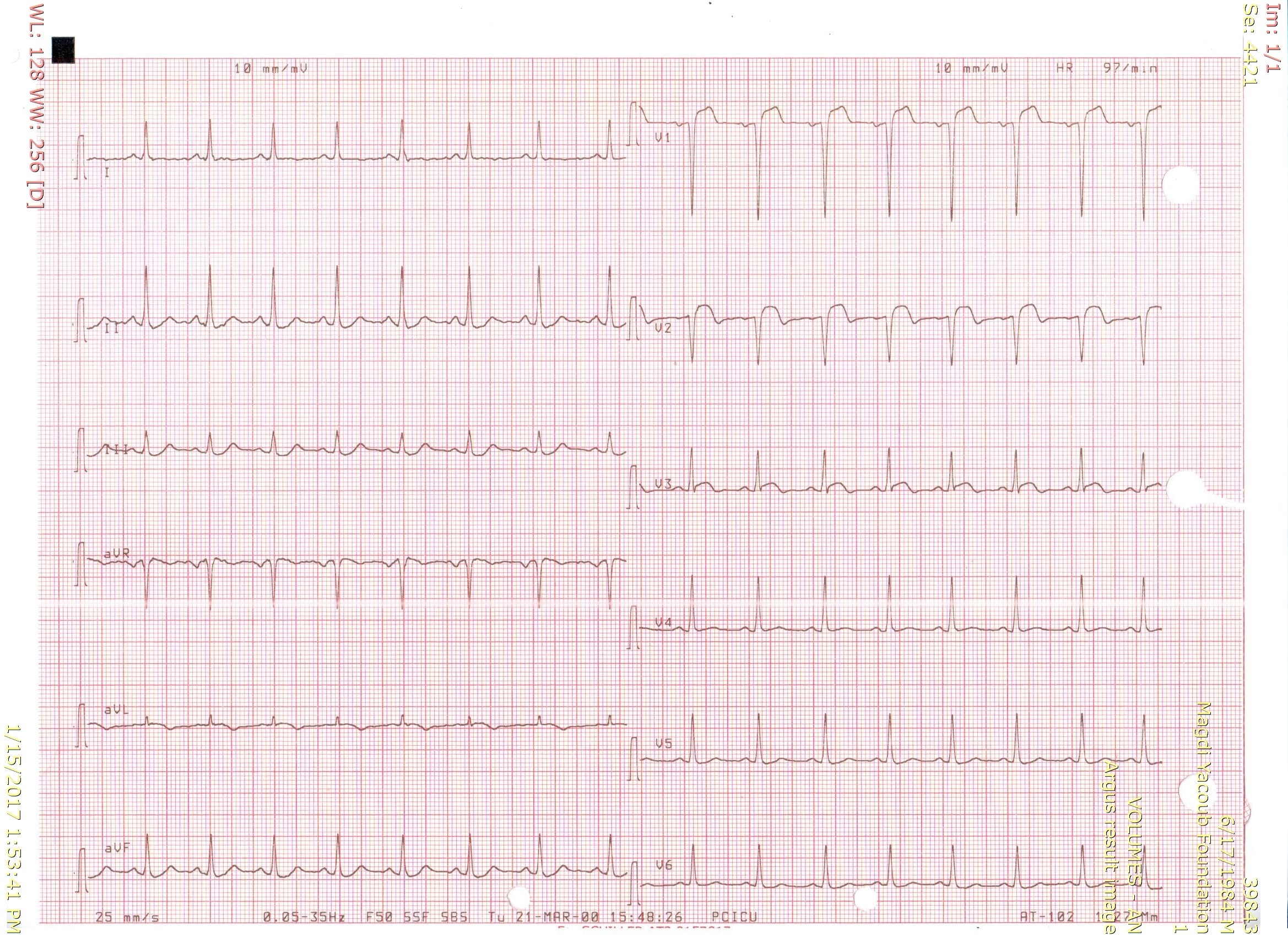
Figure 1. Twelve lead surface ECG showing pathological Q waves in leads V1 & V2, ST segment elevation in leads V1 to V3 and reciprocal ST segment depression in inferior leads.
A diagnosis of recent antero-septal ST elevation myocardial infarction (STEMI) was made and primary PCI was performed for total left anterior descending coronary artery (LAD) occlusion with a drug eluting stent (Figure 2).
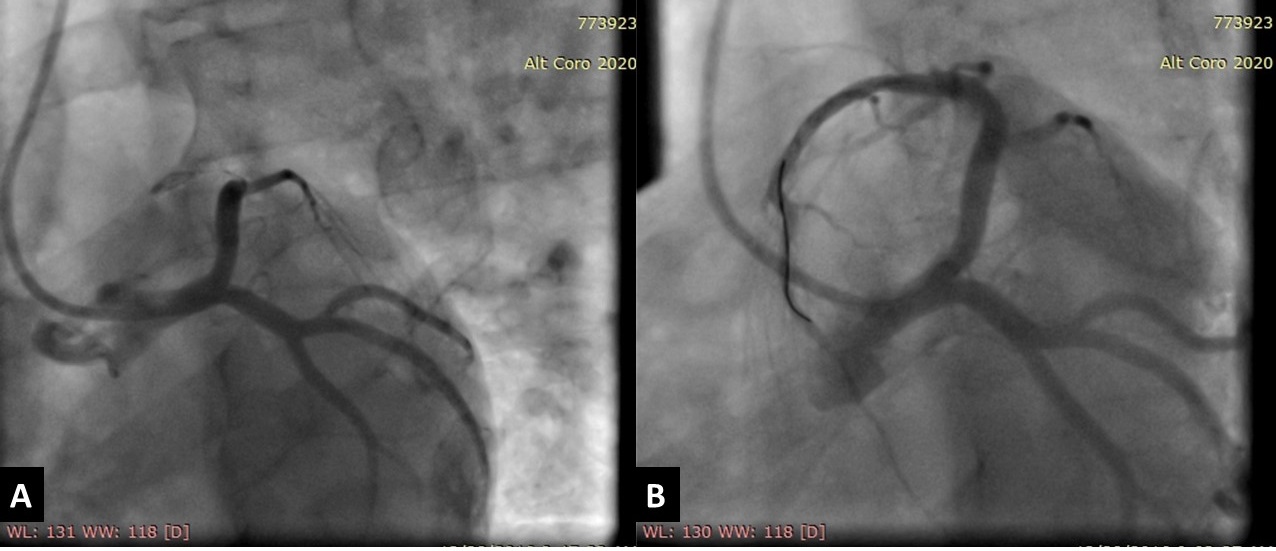
Figure 2. Invasive coronary angiography showing: (A) mid segment total occlusion of the left anterior descending artery (LAD) after giving the first diagonal branch. (B) After a successful primary percutaneous coronary intervention for the total LAD lesion with a drug eluting stent with final TIMI III flow and good angiographic results.
On the second post-procedure day, transthoracic echocardiography showed antero-septal wall hypokinesia and unexplained asymmetrical septal hypertrophy. The end-diastolic (ED) interventricular septum (IVS) wall thickness was 18.8 mm (Figure 3 and Movie 1) with no left ventricular outflow tract (LVOT) obstruction (Peak velocity across the LVOT = 1.8 m/s). For improved myocardial mapping and tissue characterization, cardiac magnetic resonance (CMR) was performed on 1.5 T scanner (Siemens Magnetom Aera, Siemens Medical Systems, Erlangen, Germany).
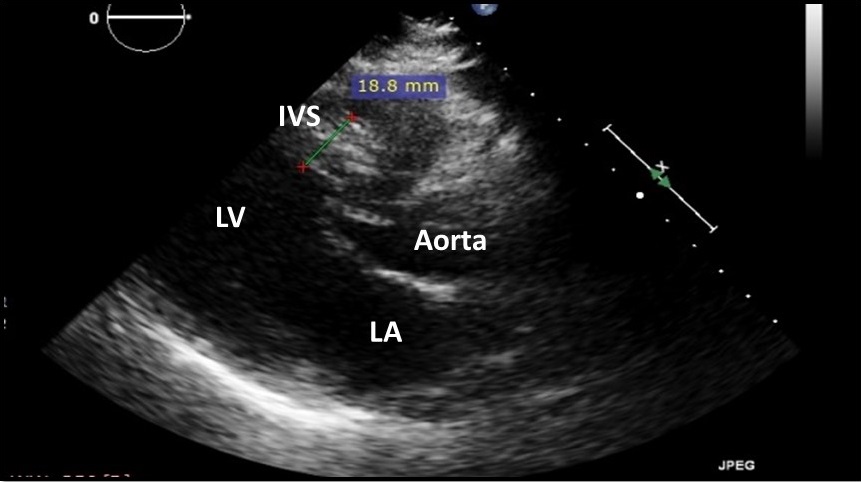
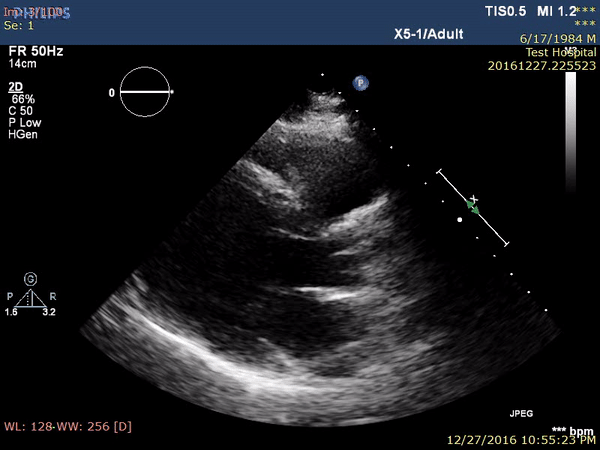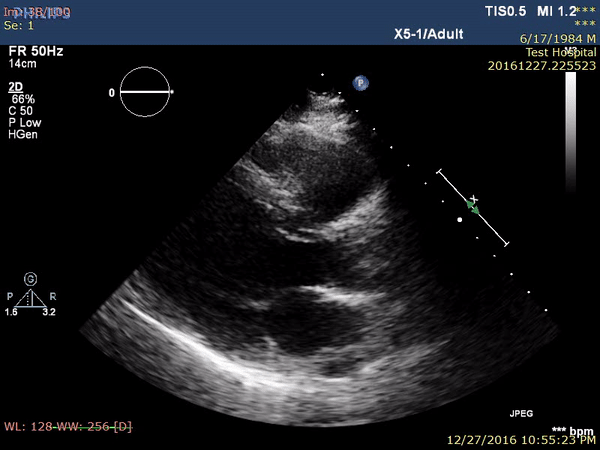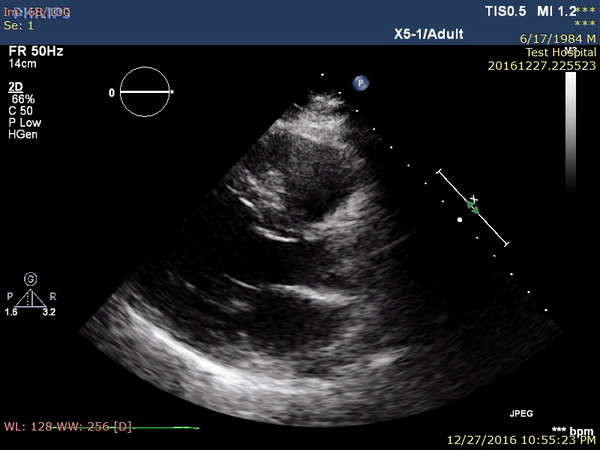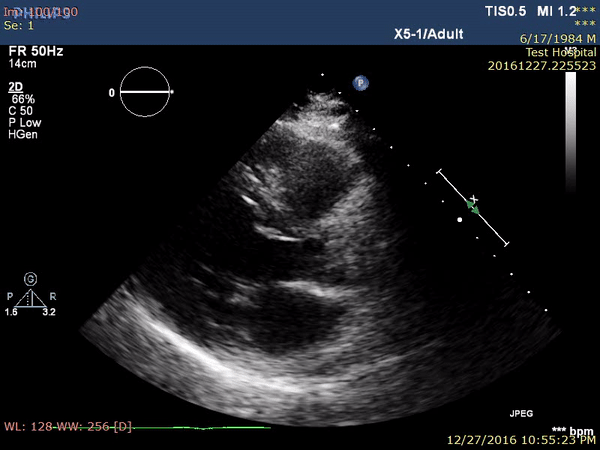
Figure 3 and Movie 1. Transthoracic Echocardiography, parasternal long axis view showing antero-septal wall hypokinesia and unexplained asymmetrical septal hypertrophy with end-diastolic (ED) interventricular septum (IVS) wall thickness = 18.8 mm and no left ventricular outflow tract (LVOT) obstruction. LV: left ventricle, LA: left atrium, IVS: interventricular septum.
CMR technique
SSFP CINE images were obtained for functional assessment, identification of LV resting wall motion abnormalities and detection of hypertrophied segments. Black blood TIRM images were obtained for assessment of water content to differentiate edematous tissues from other tissues. The patient was then injected with 0.2 ml/kg of gadolinium-based contrast agent and early gadolinium enhancement (GE) images were obtained for identification of LV thrombosis or microvascular obstruction (MVO) and delayed GE images for identification of LV fibrosis.
CMR findings
Similar to the echocardiographic findings, CMR showed thickened hypokinetic antero-septal wall with no signs of LVOT obstruction on cine SSFP images (Movie 2).
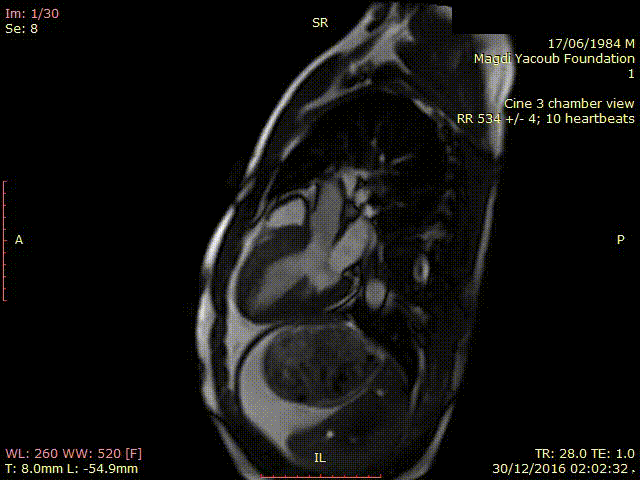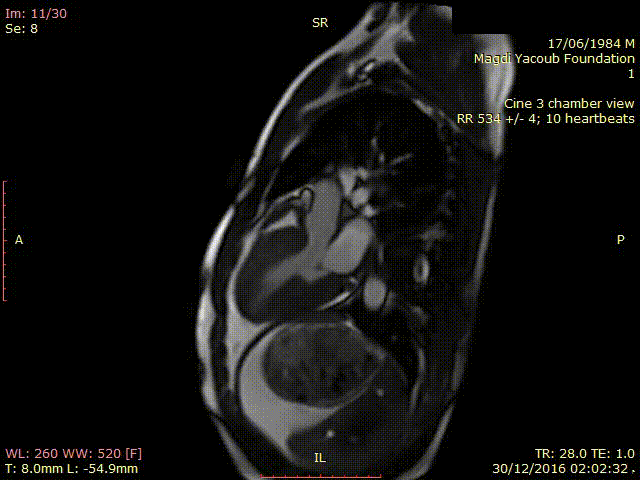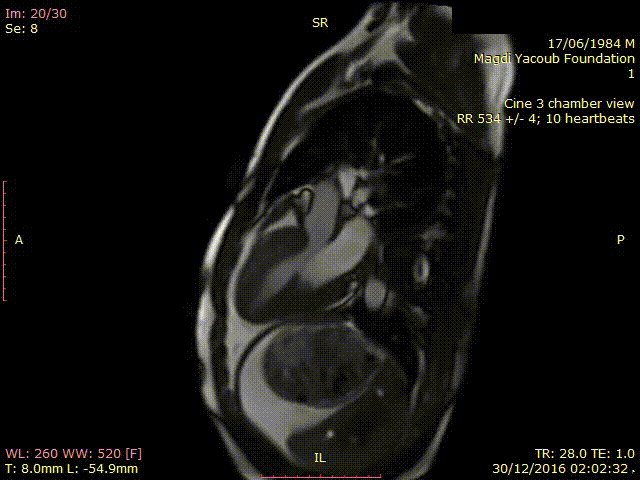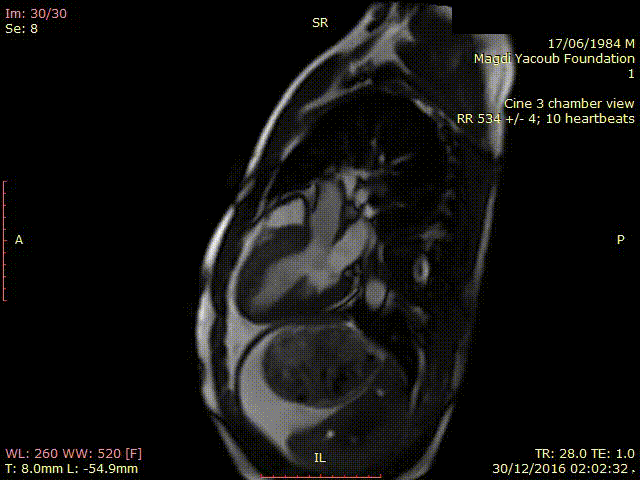
Movie 2. SSFP Cine 3 chamber view showing thickened hypokinetic antero-septal wall with no signs of LVOT obstruction.
Black blood TIRM images showed high signal intensity denoting marked myocardial edema in antero-septal wall (Figure 4). Early GE images showed large low signal area of MVO along the whole antero-septal wall (Figure 5) with the same low signal area within an enhancing site of MI on late GE images (Figure 6).
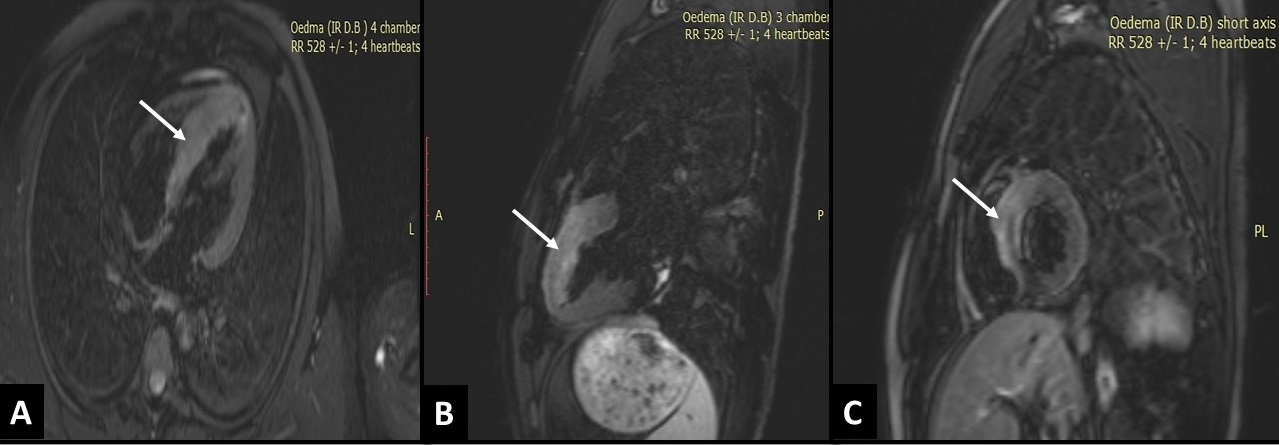
Figure 4. Black blood TIRM images in 4 chamber view (A), 3 chamber view (B) and short axis view (C) showing high signal intensity involving the antero-septal and infero-septal walls denoting marked myocardial oedema in these walls (Arrows).
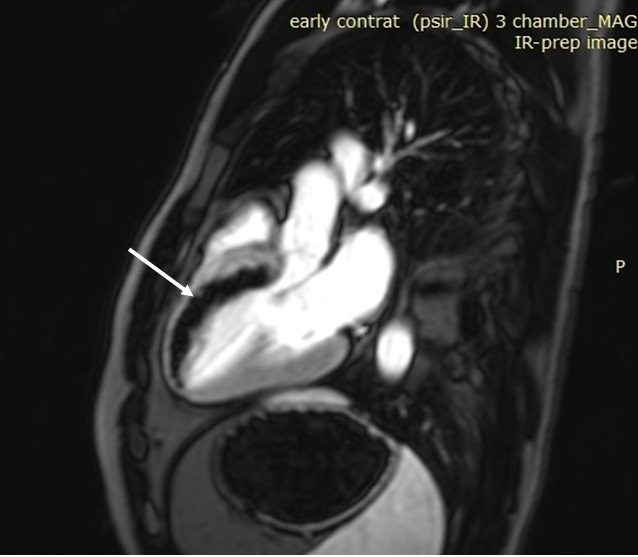
Figure 5. Early gadolinium enhancement image in 3 chamber view showing a large low signal area of microvascular obstruction (MVO) along the antero-septal wall from base through apex (Arrow).
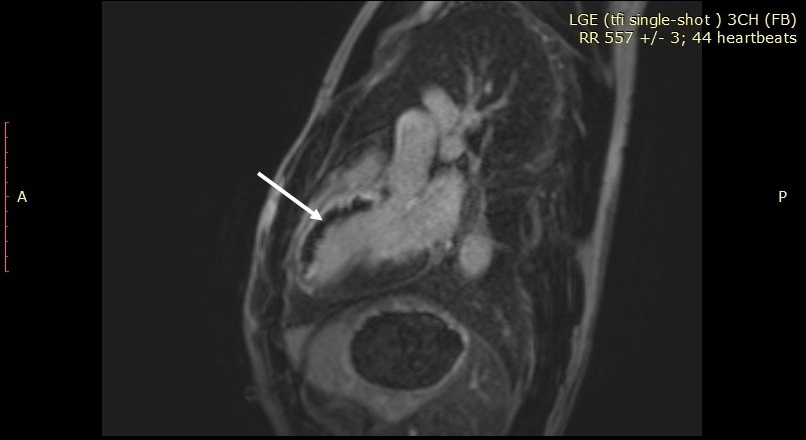
Figure 6. Delayed gadolinium enhancement image in 3 chamber view showing the same low signal area of MVO within an enhancing site of myocardial infarction involving the antero-septal wall from base through apex (Arrow).
Four months later, follow up CMR showed thinned out hypokinetic antero-septal wall on cine SSFP images (Movie 3), marked reduction of myocardial edema on black blood TIRM images (Figure 7), and transmural myocardial enhancement along the antero-septal wall on late GE images (Figure 8).
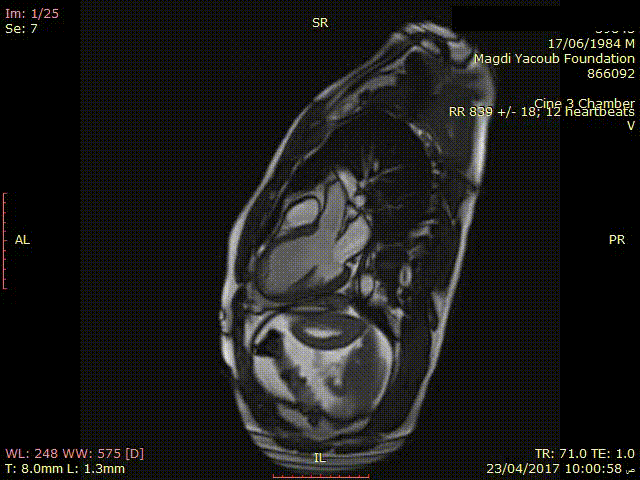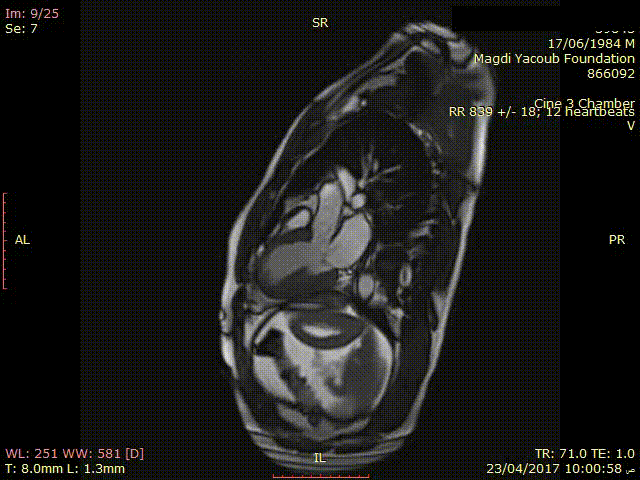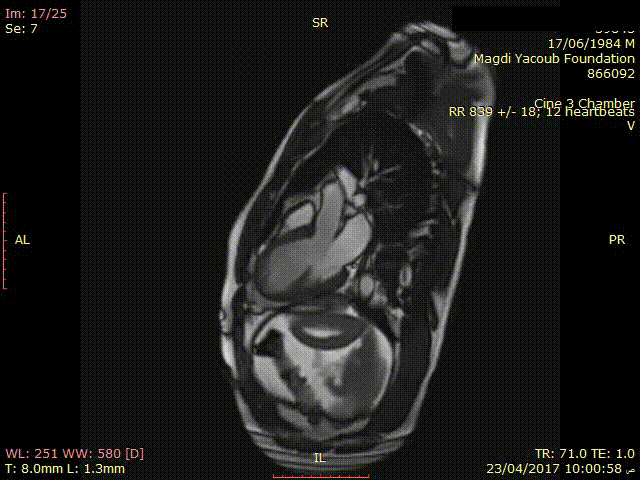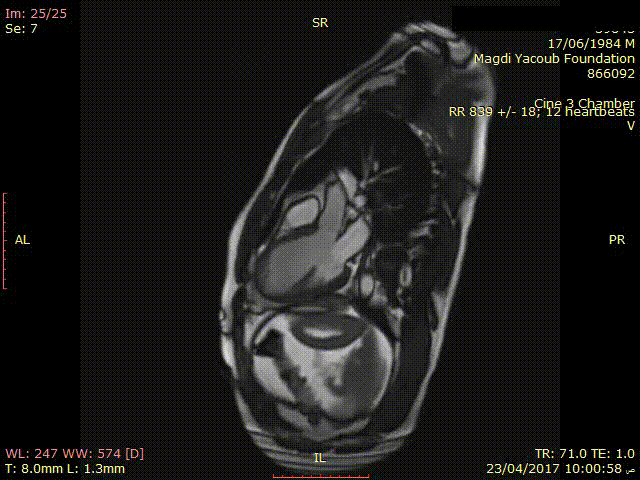
Movie 3. SSFP Cine 3 chamber view showing thinned out and hypokinetic antero-septal wall.
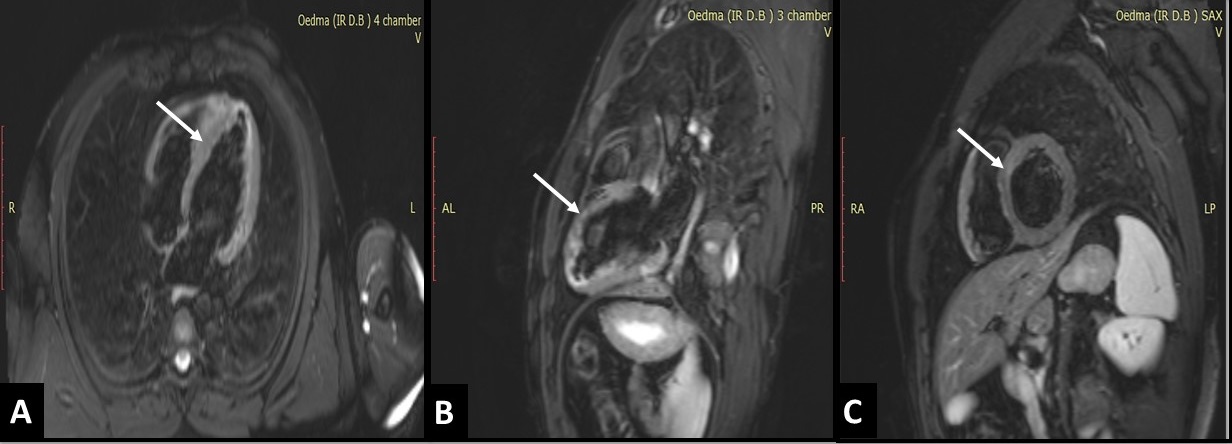
Figure 7. Black blood TIRM images in 4 chamber view (A), 3 chamber view (B) and short axis view (C) showing marked reduction of myocardial oedema in both antero-septal and infero-septal walls (Arrows).
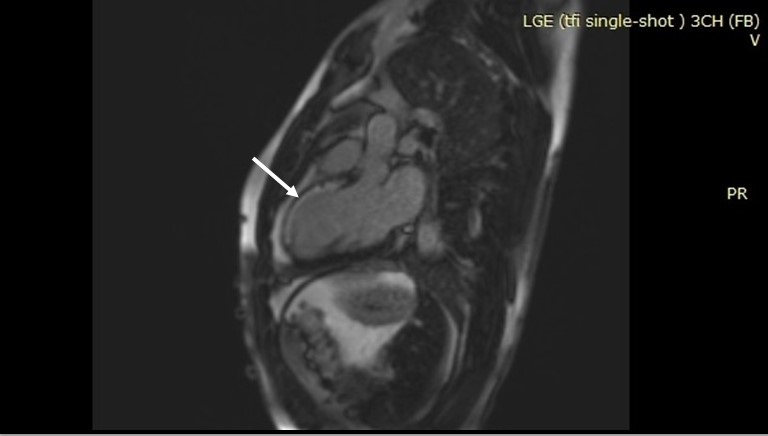
Figure 8. Delayed gadolinium enhancement image in 3 chamber view showing transmural myocardial fibrosis involving the antero-septal wall from base through apex (Arrow).
Conclusion
CMR is the diagnostic imaging modality of choice for myocardium tissue characterization that provides very important information in different cardiac pathologies which leads to better understanding of difficult cardiac abnormalities. In this case, CMR revealed that thickened IVS was due to a swollen septum by edema and MVO rather than a hypertrophied septum that occurs in cases of hypertrophic cardiomyopathy. So, in our case, CMR identified what we can call, a pseudo-HCM.
Perspective
Acute coronary syndromes (ACS) results from either partial or complete occlusion of the epicardial coronary artery. As delivery of oxygen to myocardium impairs, cellular membrane instability occurs resulting in influx of sodium ions due to ischemia-activation of the sodium-hydrogen exchange. This leads to intracellular water increase and that’s the pathogenesis behind myocardial edema development in ACS 1. Persistence of ischemia results in irreversible myocardial injury and necrosis.
As the coronary artery re-canalizes, plaque debris and thrombotic material may embolize downstream and result in occlusion of myocardial microcirculation producing the so-called ‘micro vascular obstruction. (MVO)2
Although our patient had successful revascularization and TIMI III flow, the presence of a large area of MVO denotes a “no-reflow” phenomenon which refers to a state of myocardial tissue hypoperfusion in the presence of a patent epicardial coronary artery. In this setting, CMR offers the cardiologist a comprehensive non-invasive imaging view at the tissue level that can clearly delineate the difference between myocardial edema, myocardial scar and MVO using different imaging sequences 3.
T2-weighted imaging can locate the extent of myocardial edema and area at risk following ACS, well known late gadolinium enhancement imaging is the gold standard to evaluate myocardial scarring and early gadolinium enhancement imaging with longer inversion time (TI) is recommended in the assessment of MVO.
An important differential diagnosis of this low signal area is to differentiate MVO from laminar thrombus. A low signal area seen on LGE imaging within the cavity abutting the wall of the myocardium would be consistent with thrombus4 while in our patient, the low signal was confined within the myocardium denoting a MVO rather than a thrombus.
Definite tissue characterization in ACS has a major clinical implication on disease progression and prognosis either in detecting area at risk and salvageable myocardium or in detecting areas of MVO which carry poor prognosis and correlate closely with adverse LV remodeling. Besides, specifically in our case, CMR is useful in differentiation between myocardial edema and other causes of myocardial hypertrophy.
References
2. Walls MC, Verhaert D, Min JK, Raman S V. Myocardial edema imaging in acute coronary syndromes. J Magn Reson Imaging. 2011;34(6):1243-1250. doi:10.1002/jmri.22737.
3. Bogaert J, Eitel I. Role of cardiovascular magnetic resonance in acute coronary syndrome. Global Cardiology Science & Practice. 2015;2015(2):24. doi:10.5339/gcsp.2015.24.
Case prepared by Madhusudan Ganigara, MD.
Link to CloudCMR-





