Sameh Khalil, MD, Sara Ragab, BSc, Omnia Mohammed BSc, Taher Moustafa BSc, Ahmed Hamdy, BSc, Shimaa Ahmed, MD, Ahmed Heny, MD and Maha Taher, MD.
Alfa Scan Radiology Center, Cardiovascular Imaging Department, Cairo, Egypt
Case Published in the Journal of Cardiovascular Magnetic Resonance: Click here for the link
Click here for PubMed Reference to Cite this Case
Clinical History
A 43-year-old male patient underwent left side radical nephrectomy four months ago for removal of biopsy proven clear renal cell carcinoma. The patient did not receive chemotherapy. The patient presented with complaints of shortness of breath and hemoptysis. Echocardiogram revealed a left atrial mass with no right heart involvement detected. As renal cell carcinomas most commonly involve the heart by extension through the IVC and right-side chambers, a left atrial thrombus was initially suspected in this case. The patient was referred for cardiac MRI to verify the diagnosis prior to starting anticoagulants.
CMR Findings
CMR was performed using a 1.5 T scanner (Siemens Magnetom Aera, Siemens Medical Systems, Erlangen, Germany). Given the patient’s shortness of breath, real time axial cine and black blood axial images were acquired through the chest and upper abdomen. These images revealed widespread metastatic masses involving the liver, mediastinum and lungs with extension through the pulmonary veins. The mediastinal masses appeared continuous with the left atrial mass, raising the possibility of extension of the malignant tumor to the left atrium.
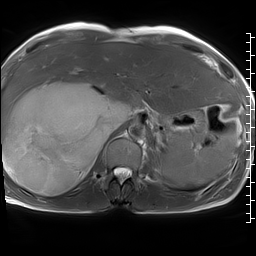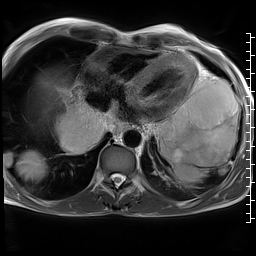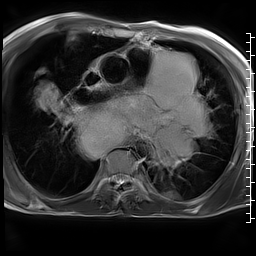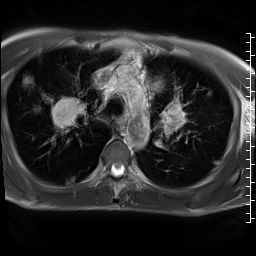
Video 1: Axial black blood stack demonstrating widespread masses involving the chest and abdomen
For mass characterization, ECG gated axial T2, axial T2 with fat suppression, axial T1, and first pass contrast imaging after injection of a gadolinium-based contrast agent (Magnevist, Schering AG, Berlin, Germany, 0.2 mmol/kg), 1, 5 and 10 minutes after contrast injection images were acquired. ECG gated delayed post contrast images were acquired with high inversion time (700 msec) to assess for possible thrombus.
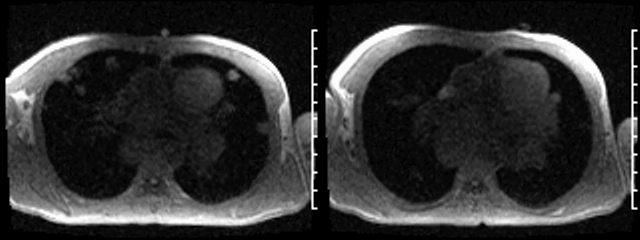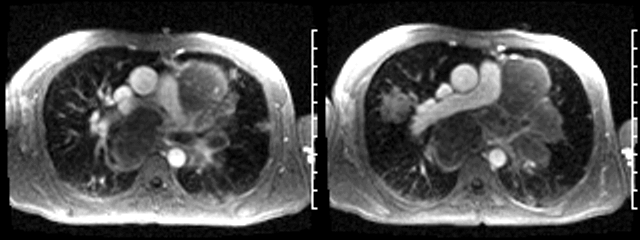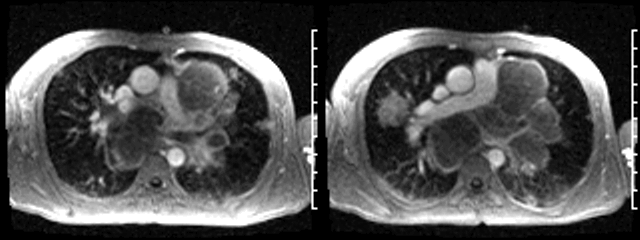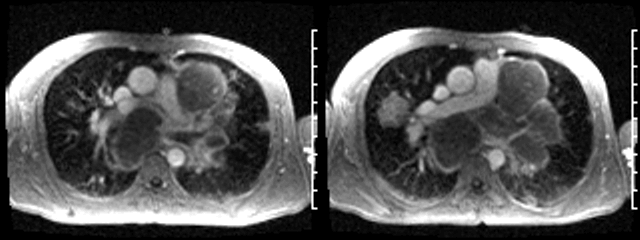
A single, well defined, infiltrative mass was seen in the left atrium measuring 5.7 x 5.3 x 3.6 cm (Video 2). The atrial mass compromised mitral valve inflow, protruding into the left ventricle. No other cardiac chamber or valvular involvement was detected. The mass extended into the left atrium through infiltration of the left sided pulmonary veins, a part of multiple mediastinal and bilateral lung metastases (Figure 1).
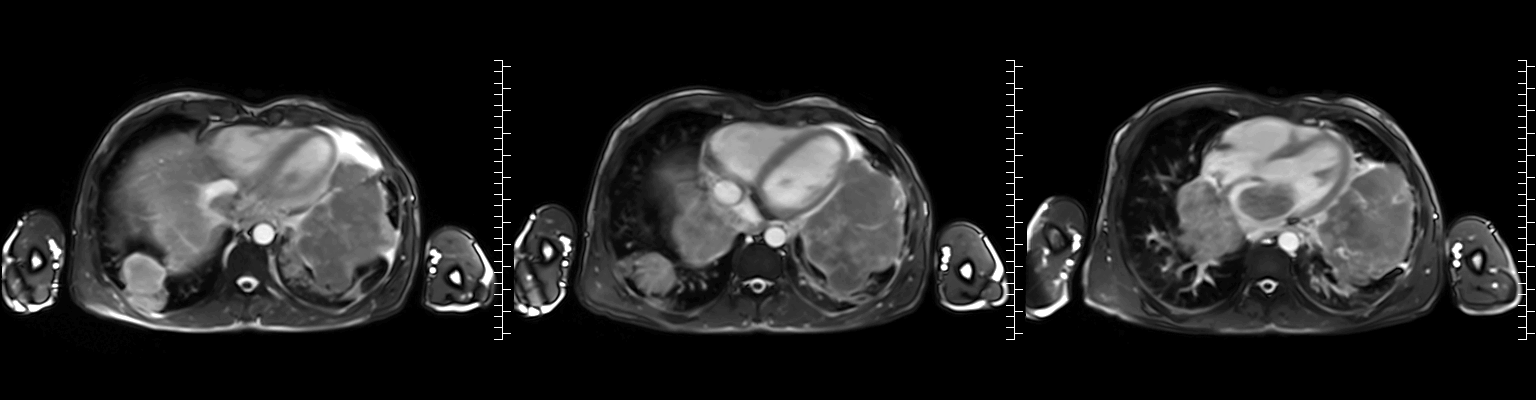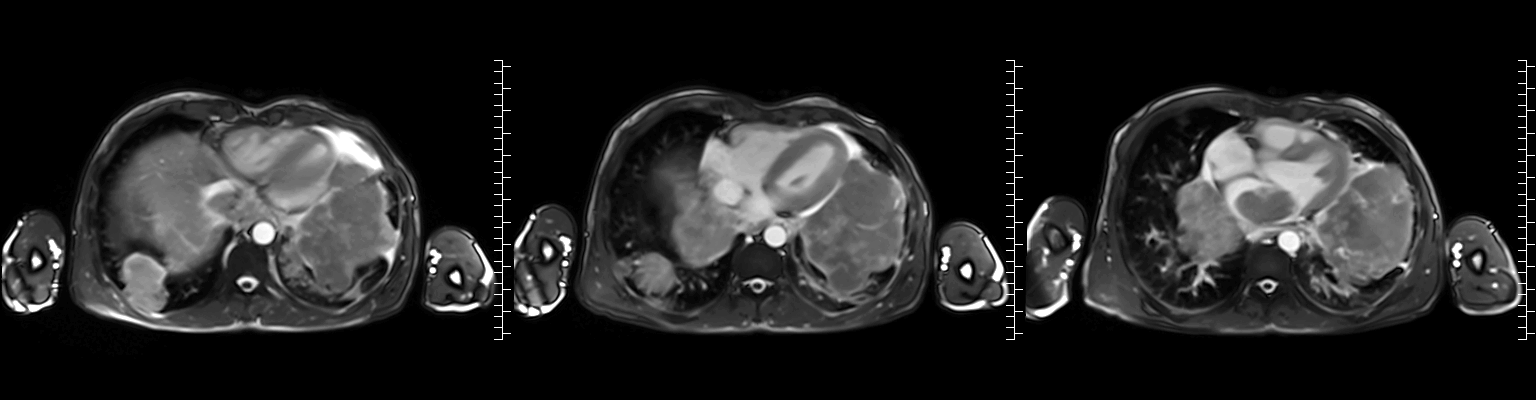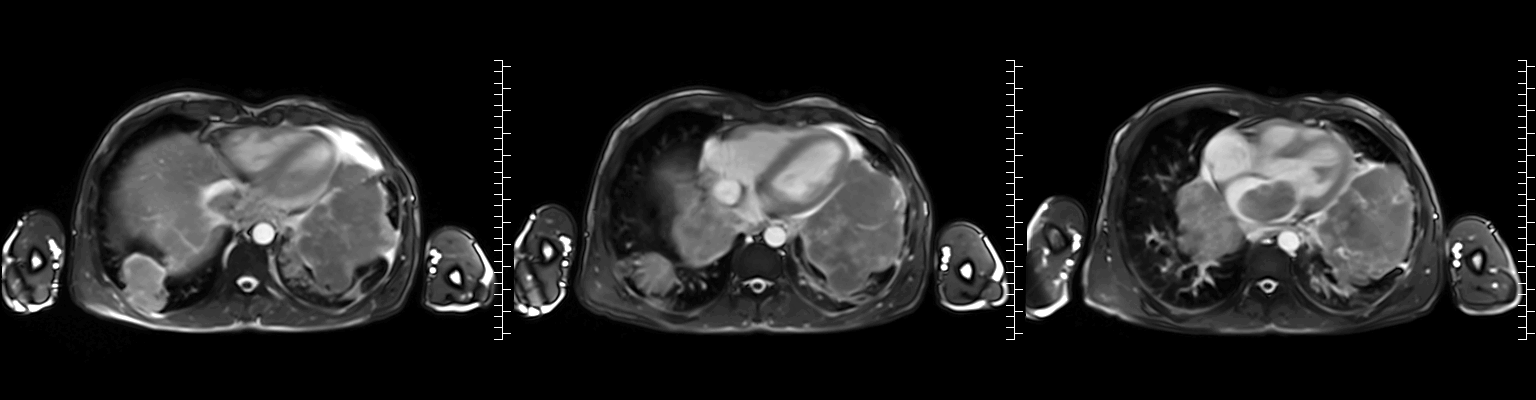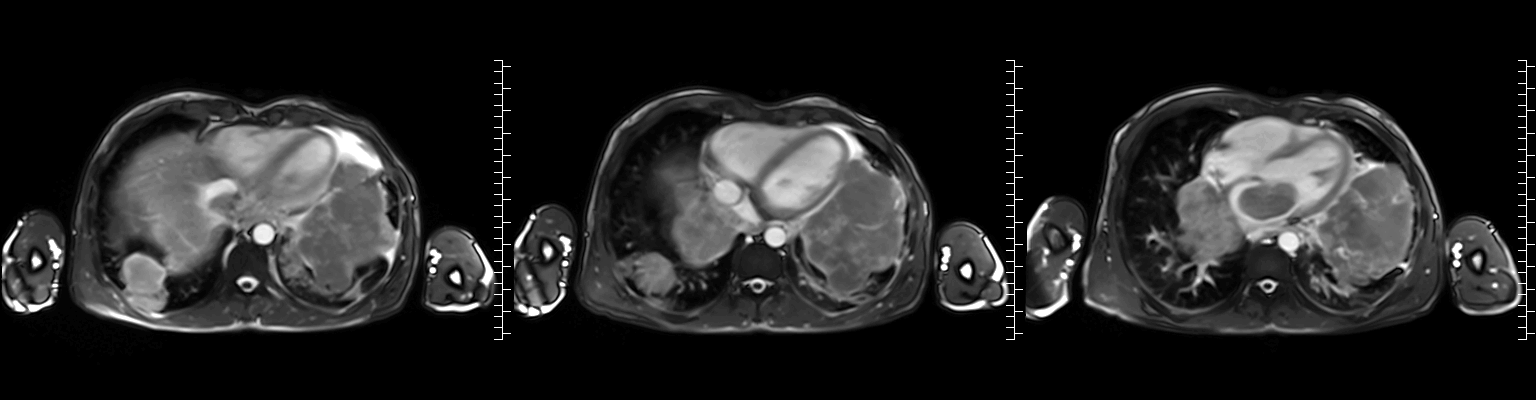
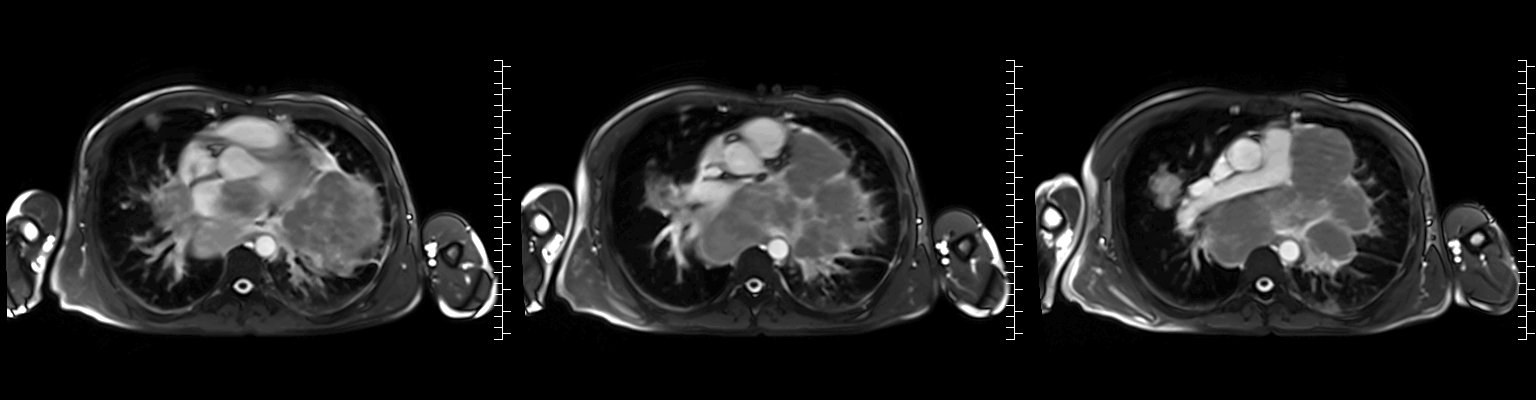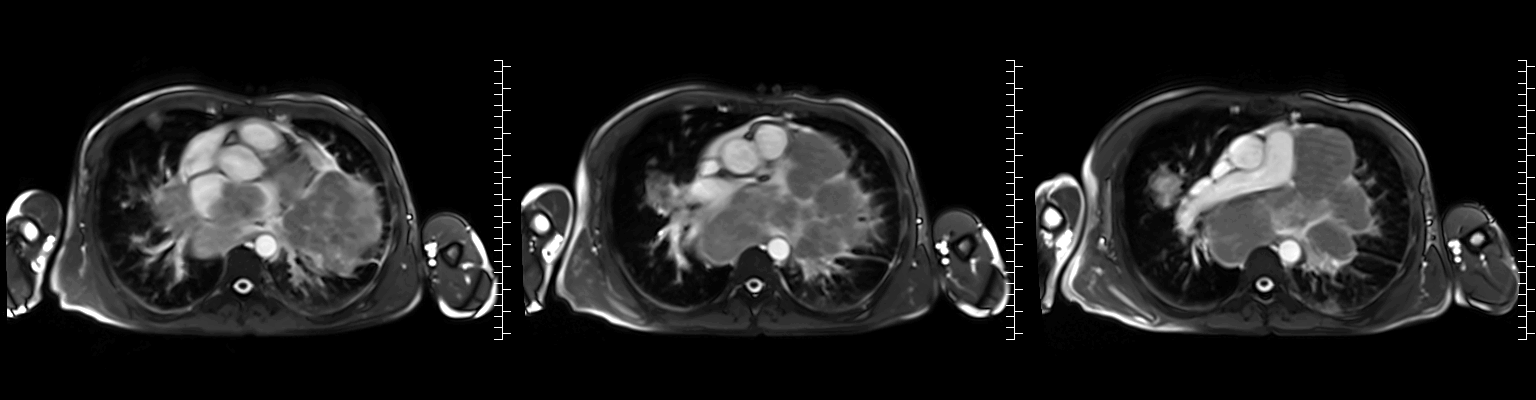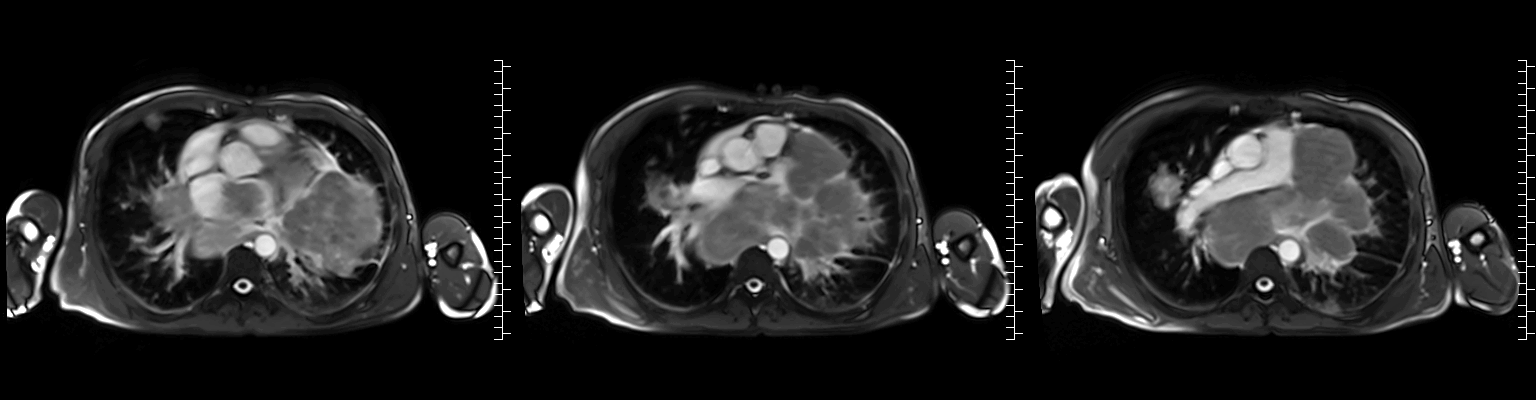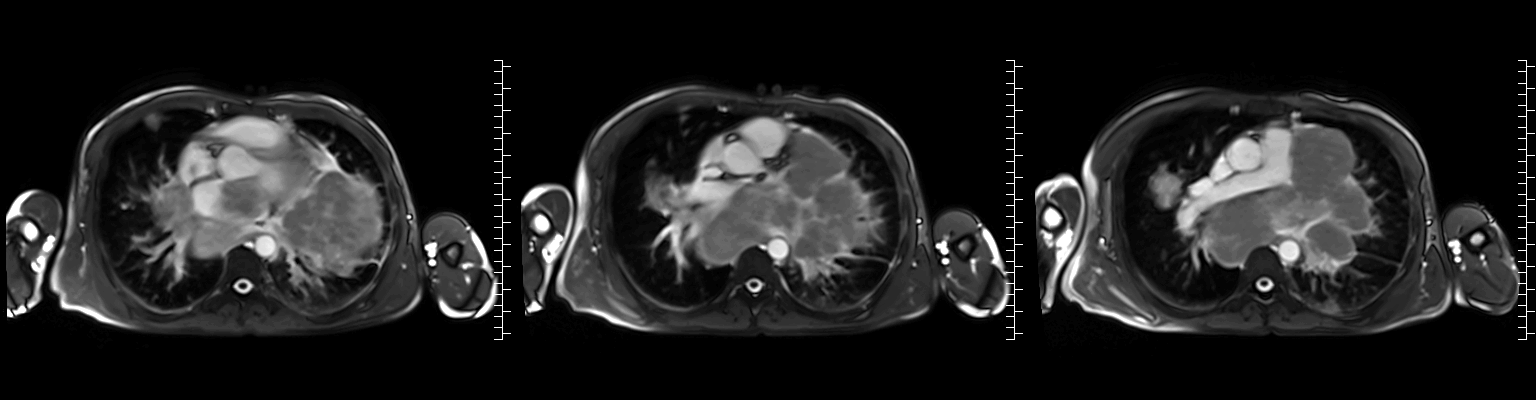
Video 2: Axial cine stack (multiple slice planes, inferior to superior) demonstrating the complex mediastinal mass infiltrating the left pulmonary veins, extending to the left atrium and compromising mitral valve flow; remaining cardiac chambers are free from infiltration.
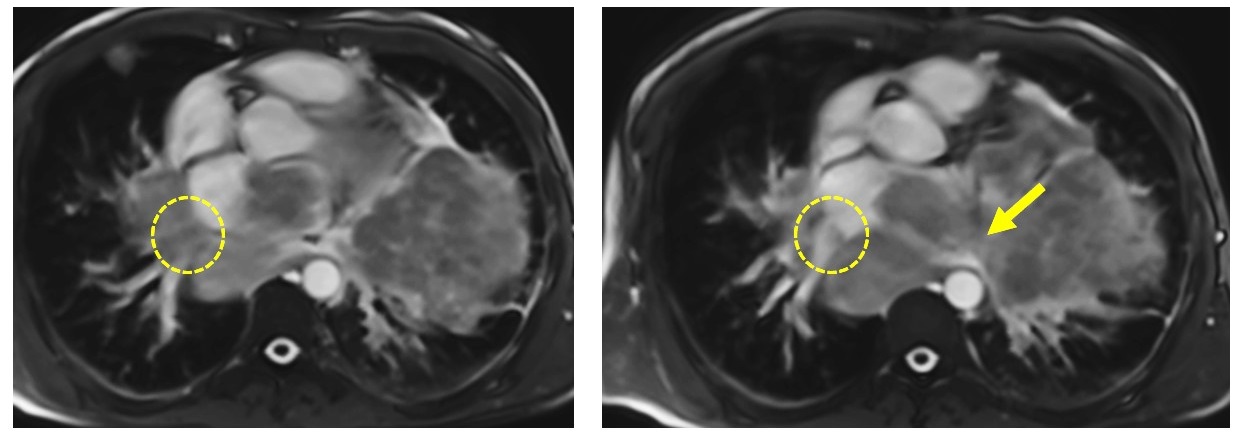
Figure 1: ECG-gated cine images show the right pulmonary veins (yellow circle) and left pulmonary veins (yellow arrow) infiltrated by the mediastinal mass.
Biventricular size and systolic function were normal. Focal pericardial infiltration over the left ventricle and along the posterior and inferior pericardial surfaces was noted (Figure 2). Multiple pleural masses were also seen. No pericardial or pleural effusions were present.
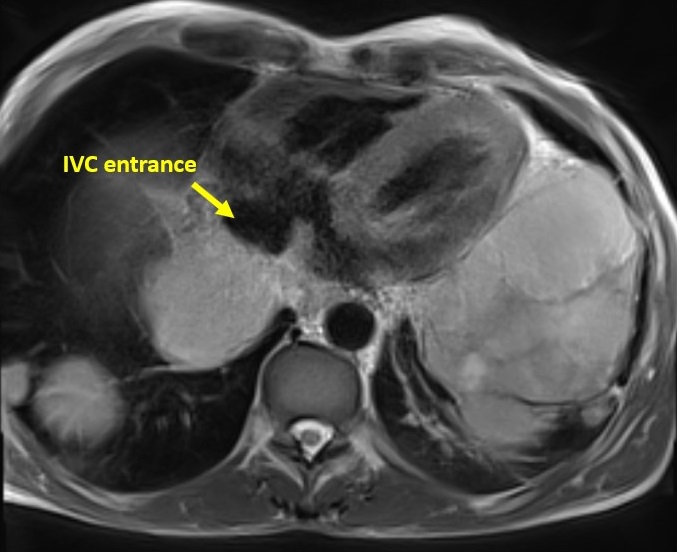
Figure 2: ECG-gated T2WI fat suppression axial image shows pericardial infiltration over the atria, yet, no effusion and no atrial extension. Also, the IVC entrance to the right atrium is spared.
The mass demonstrated hyperintense T1 and T2 signal relative to the myocardium
(Figures 3 and 4).
Figure 3: T2 weighted axial image shows a large mediastinal metastatic mass (yellow arrows) infiltrating the left atrium (LA) and encasing the main pulmonary artery (MPA). The mass appears separable from the ascending aorta (AO). The right lower pulmonary vein (PV) appears infiltrated by mass (red arrow).
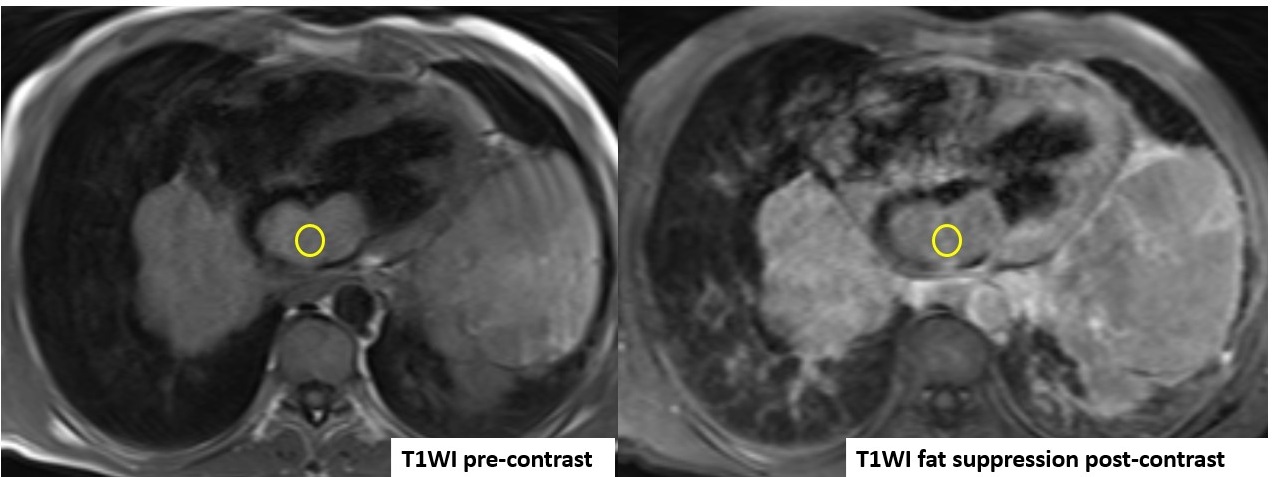
Figure 4: ECG-gated T1WI before and after contrast, with increased signal intensity in the post-contrast study relative to the pre-contrast study.
Mild enhancement was noted during first pass perfusion (Video 3), with mild enhancement also seen in early post contrast images. This could be secondary to contrast moving over the surface of the mass as opposed to true mass perfusion. Decreased delayed enhancement was detected (Figure 5). There was a possible hemorrhagic component. No calcifications, central breakdown, or fatty components to the mass were detected.
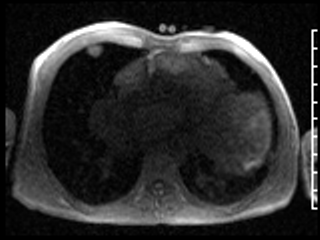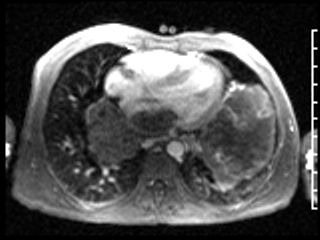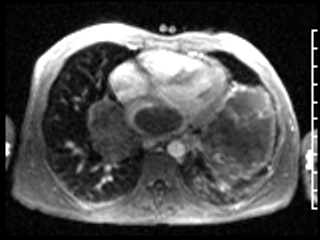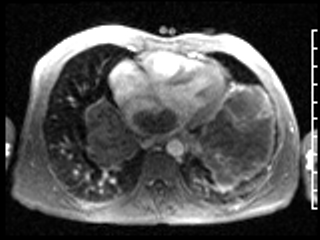
Video 3: ECG-gated first pass perfusion study shows hypovascular nature of the tumor with faint enhancement
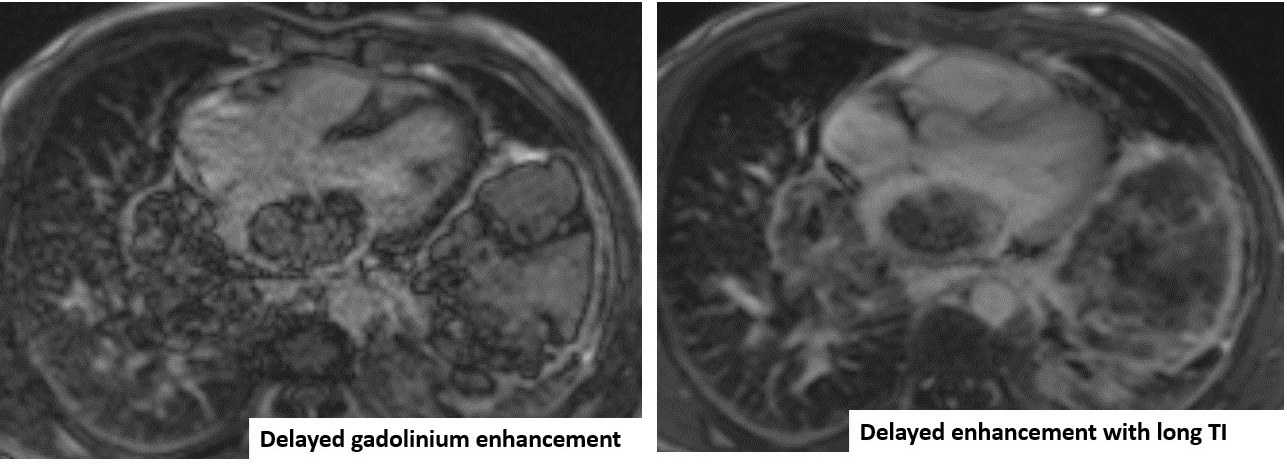
Figure 5: ECG-gated delayed gadolinium enhancement. In comparison to the long TI image, there is still enhancement in the mass with no jet-black signal seen in the long TI image.
Regarding the great vessels, the thoracic aorta was spared from metastatic involvement, with the arch and descending thoracic aorta passing tangential to the mediastinal masses with no definite intravascular extension or narrowing of the arch detected. The left pulmonary artery is infiltrated with attenuation of its diameter. No definite intravascular involvement was detected in the main or right pulmonary arteries (Video 4).
Video 4: First pass perfusion sequence demonstrates infiltration of the left pulmonary artery.
There is a very large liver mass measuring 14 x 11 cm seen infiltrating the right sided hepatic veins and compressing the hepatic portion of IVC (Figure 6). There is absent connections between the extra-thoracic and hepatic vein malignant tissue and the intra-cardiac tumor. While the hepatic portion of the IVC was spared, wall infiltration with narrowing was seen just before entrance into the right atrium.
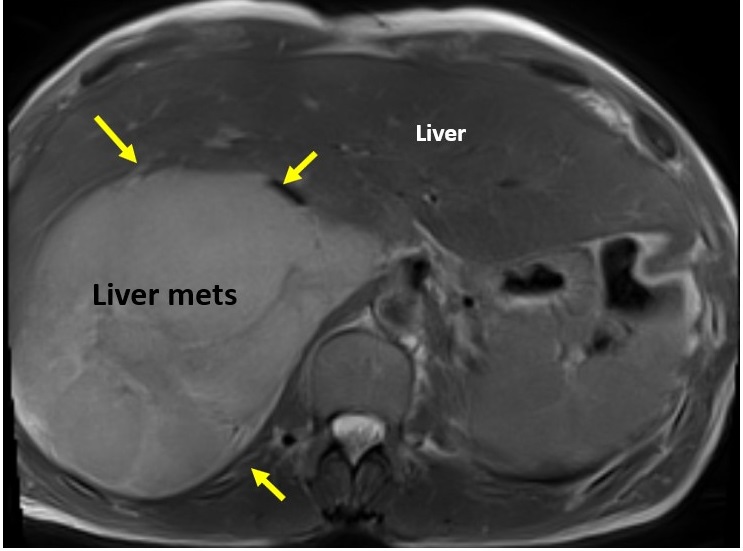
Figure 6: Large liver mass indicated by yellow arrows.
Multiple metastases were seen in the lungs bilaterally, with largest lesion seen in the left lower lung lobe measuring 10 x 7.5 cm. Multiple mediastinal and para-hilar lymph nodes were seen, the largest of which was in the subcarinal group measuring 4.7 cm.
Conclusion
While aggressive chemotherapy and thrombolytics were intially planned, the plan was altered due to the CMR findings. Based on the CMR findings, the masses visualized were felt to most likely be consistent with widespread metastatic disease. Ultimately, palliative chemotherapy was offered and the patient unfortunately died. Pathology assessment of the cardiac and mediastinal masses was not performed.
This case highlights the importance of an organized approach regarding clinical decision making and diagnostic imaging studies performed, as management approaches can sometimes be changed based on confirmatory testing.
Perspective
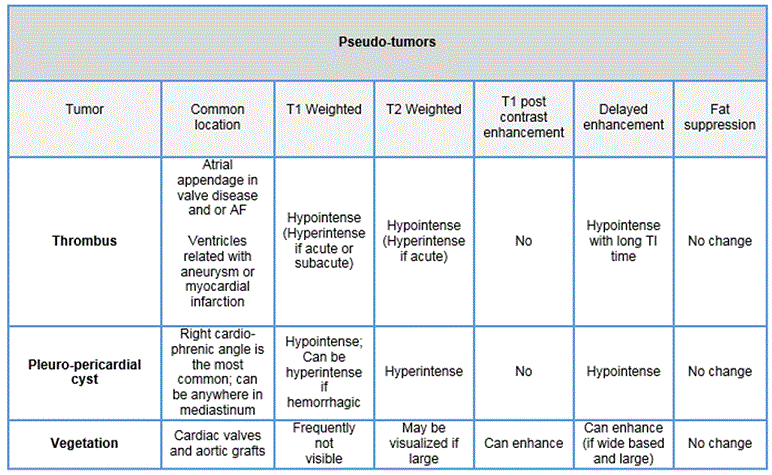
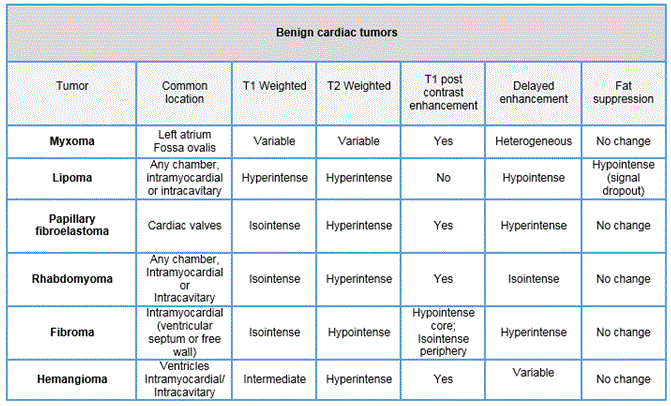
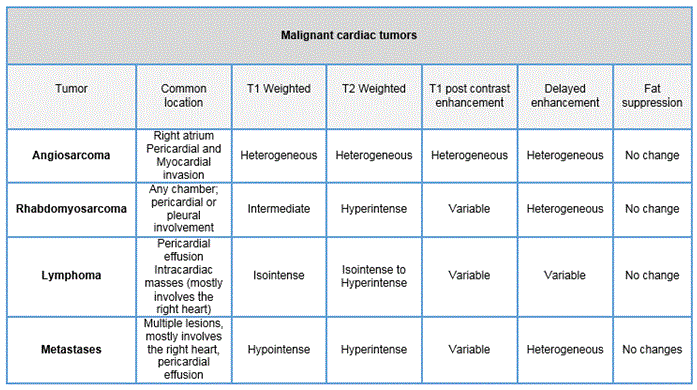
Cardiovascular magnetic resonance imaging is an invaluable noninvasive imaging modality to assess cardiac masses. Tables 1-3 below provides a summary of the MR imaging characteristics seen with various types of cardiac masses (1).
Renal cell carcinoma patients remain at risk for metastatic disease for many years after diagnosis, with the clear cell type having the highest risk for metastatic spread (2). Cardiac metastasis of renal cell carcinoma is an exceptional event, particularly when there is lack of inferior vena cava involvement (3).
To our knowledge, only 14 cases are reported worldwide involving the pulmonary veins and or the left atrium without inferior vena cava infiltration (4-6). Absent extra-renal metastatic spread before radical nephrectomy, absent inferior vena cava infiltration, and atypical route of metastases all were unusual findings in this case.
Click here to view the entire CMR study on Cloud CMR.
References
3. Kojiro Ohba, Yasuyoshi Miyata, Kensuke Mitsunari, Tomohiro Matsuo, Yasushi Mochizuki and Hideki Sakai. Left atrial metastasis of renal cell carcinoma: a case report and review of the literature. BMC Research Notes 2014, 7:520.
4. Cochennec F, Seguin A, Riquet M, Fabiani JN. Intracardiac renal cell carcinoma metastasis. Eur J Cardiothorac Surg. 2008; 34(3):697-699.
Associate Editor: Robert D. Tunks, MD







