Rashid Al Umairi, MD, FRCR, FSCMR
Faiza AL Kindi, MD
The Royal Hospital
Muscat, Oman
Case Published in the Journal of Cardiovascular Magnetic Resonance: Click here for the link
Click here for PubMed Reference to Cite this Case
Clinical History: A 59-year-old man, admitted with a history of non-ST-elevation myocardial infarction and treated with percutaneous coronary intervention (PCI), underwent echocardiogram and found to have a mass attached to the septal leaflet of the tricuspid valve suggestive of a papillary fibroelastoma. Cardiovascular MRI was requested to exlude other differential diagnosis such as thrombus and vegetation.
CMR Findings: Four chamber cine (Movie 1, Image 1) and short axis oblique (SAO) cine (Movie 2, Image 2) confirmed the presence of an isointense mass attached to the septal leaflet of the tricuspid valve, measuring 9 x 7 mm. Delayed gadolinium enhancement (DGE) imaging demonstrated contrast enhancement throughout the mass (Image 3).
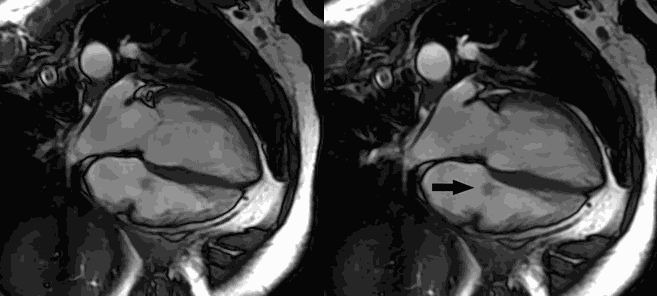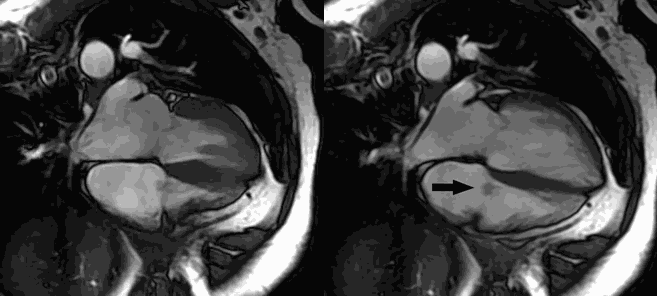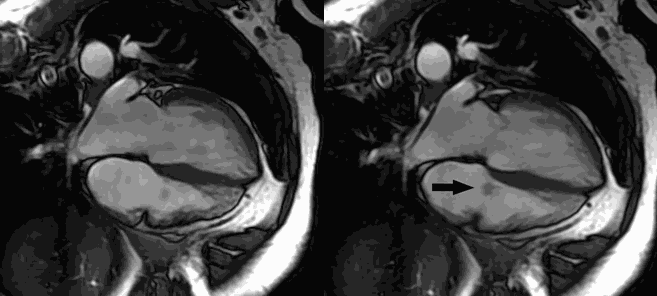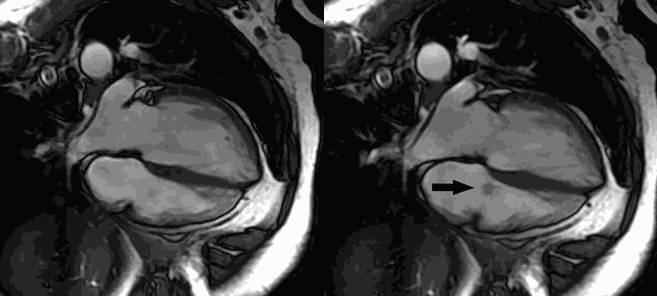
Movie 1 and Image 1: Four chamber cine SSFP with an isointense mass (arrow) on the tricuspid valve.
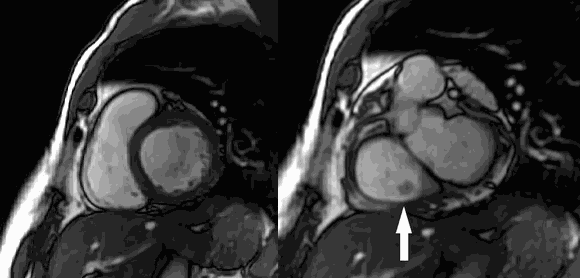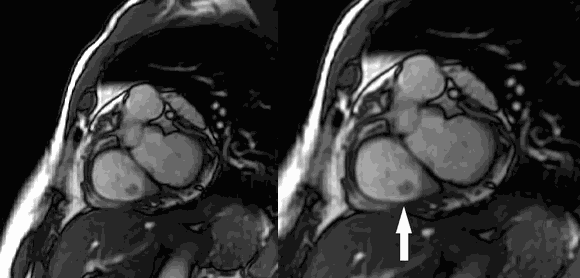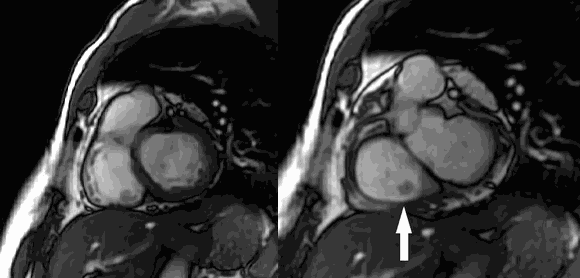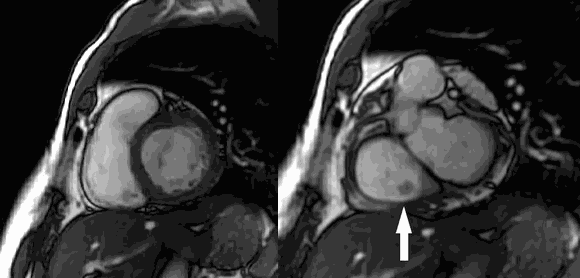
Movie 2 and Image 2: Short axis cine SSFP with an isointense mass (arrow) on the tricuspid valve.
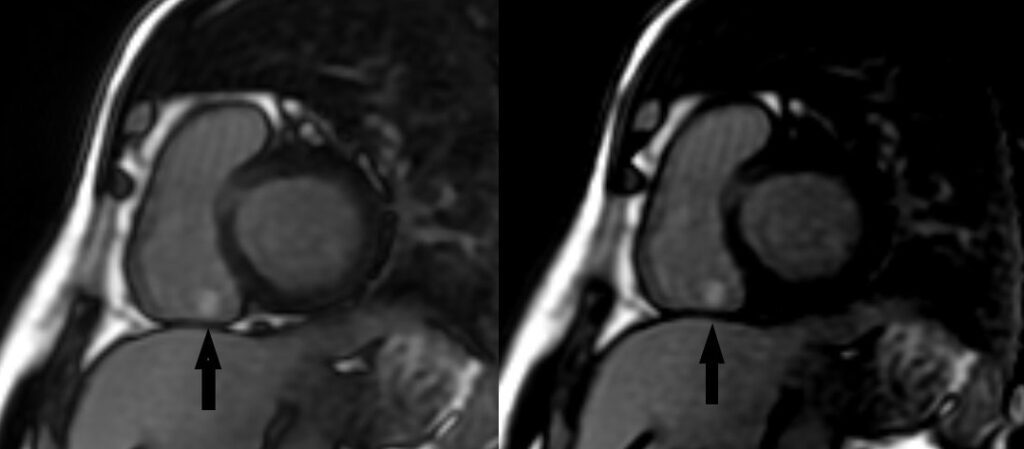
Image 3: Short axis delayed enhancement with high inversion (left) and null inversion (right) times showing a hyperintense mass (arrows) on the tricuspid valve.
Conclusion: CMR findings of the tricuspid valve mass were suggestive of a tricuspid valve papillary fibroelastoma (PFE). The patient underwent elective surgical resection and the histopathological findings were consisting with the diagnosis of PFE.
Perspective: Cardiac tumors are rare with an estimated prevalence, according to autopsy reports, of 0.002-0.3% (1). The majority of these tumors (75%) are benign and their clinical presentation is influenced by the size, location and embolism tendency (2).
PFE’s are the second most common cardiac tumor and 70-80% of valvular tumors are PFEs (3). Other valvular tumors include myxoma, nonbacterial thrombotic endocarditis, and infective vegetation (4).
The majority of PFE’s are related to the aortic valve where they usually arise from the aortic side of the aortic valve and can present with myocardial infarction due to dynamic obstruction of the coronary ostia or transit ischemic attack and strokes due to thromboembolism. The mitral and tricuspid valves are the second most common sites of involvement and the tumor usually arises at the atrial side of the valve. Pulmonary valve PFE’s are very rare with few cases reported in the literature (5).
Imaging plays an essential role in the diagnosis of cardiac PFE’s. Most cardiac PFE’s are detected by echocardiogram, transesophageal echocardiogram is more sensitive compared to transthoracic echocardiogram (6). Typically, cardiac PFE appears on echocardiogram as a rounded, oval or irregular shaped lesion with well-demarcated margin that attaches to a cardiac valve with a short mobile stalk (7). Cardiac MRI can accurately differentiate cardiac tumors from thrombus by utilizing the inversion recovery technique acquired with a prolonged inversion time (>600ms) after contrast administration. Tumors usually have intermediate signal intensity to the myocardium and the blood pool whereas thrombus and vegetations appear dark. On late gadolinium enhancement sequence, thrombus and vegetations do not enhance as they are avascular structures. On the other hand, the majority of cardiac tumors are usually hyperintense owing to their vascularity (2). Nevertheless, cardiac tumors such as lipoma, rhabdomyoma and lymphoma can show no contrast enhancement (8).
Surgical resection is usually appropriate for symptomatic patients who are candidates for surgery. However, older age, comorbid conditions and the uncertainty of the thromboembolic risk can influence the decision of surgery (6). Long term anticoagulation is recommended for symptomatic patients who are not fit for surgery however, no data strongly validate the efficacy of this approach (6). There are no clear guidelines for the management of PFE’s detected incidentally. Nevertheless, many surgeons recommend surgical resection for all left-sided PFE’s due to the protentional risk of thromboembolism PFE’s (9,10). Although extremely rare, recurrence of cardiac fibroelastoma following surgical resection has been reported (6,11,12).
Click here to view the entire study via CloudCMR.
References:
1. Leja MJ, Shah DJ, Reardon MJ. Primary cardiac tumors. Tex Heart Inst J. 2011;38(3):261-2.
Case prepared by:
Jason N. Johnson, MD MHS
Associate Editor, SCMR Case of the Week
Le Bonheur Children’s Hospital, University of Tennessee Health Science Center, St. Jude Children’s Research Hospital







